Often pelvic floor therapists see men for post-prostatectomy urinary leakage. However, at least for me, that quickly led to seeing male patients for pelvic pain and sexual dysfunction. Male sexual dysfunction is a broad category and can consist of erectile dysfunction (ED), ejaculation disorders including premature ejaculation (PE), and low libido -- often there is a pelvic floor muscle (PFM) dysfunction component. Conservative treatment frequently consists of pharmacological and lifestyle changes for this population.
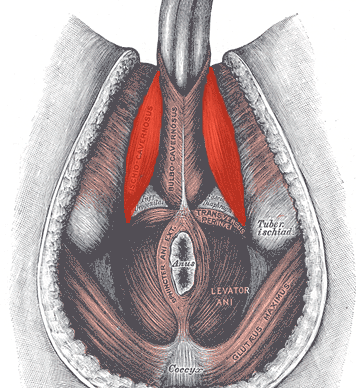
In normal sexual function, the male superficial pelvic floor musculature (bulbocavernosus and ischiocavernosus) work together to create increased intracavernosus pressure by limiting venous return, resulting in an erection. Ejaculation is created by rhythmic contractions of the bulbocavernosus muscle.
 The authors of this systematic review were curious if pelvic floor muscle training was effective for treating erectile dysfunction and premature ejaculation diagnoses, and if so to determine whether there is a treatment protocol. Ten studies were found that met the inclusion criteria, five that focused on ED and five that focused on PE. In total, there were 668 participants ranging in age from 30-59 years old. Studies were excluded if participants were post-prostatectomy and/or had a neurological diagnosis. The intervention was a pelvic floor program, and pelvic floor muscle contractions were either taught or supervised. Studies also included supportive treatment including biofeedback, lifestyle changes, and electrical stimulation.
The authors of this systematic review were curious if pelvic floor muscle training was effective for treating erectile dysfunction and premature ejaculation diagnoses, and if so to determine whether there is a treatment protocol. Ten studies were found that met the inclusion criteria, five that focused on ED and five that focused on PE. In total, there were 668 participants ranging in age from 30-59 years old. Studies were excluded if participants were post-prostatectomy and/or had a neurological diagnosis. The intervention was a pelvic floor program, and pelvic floor muscle contractions were either taught or supervised. Studies also included supportive treatment including biofeedback, lifestyle changes, and electrical stimulation.
The studies focused on erectile dysfunction listed a combination of hormonal, psychogenic, arteriogenic, and venogenic causes. The pelvic floor training ranged from 5-20 visits over 3-4 months and included a home exercise program. Pelvic floor training was similar in all studies and consisted of maximal quick contractions over one second and submaximal endurance holds over 6-10 seconds. Compliance to home exercise program was not assessed. Between 35% and 47% of participants reported a full resolution of symptoms. Subjective improvements were supported by improved maximal anal pressure and intracavernosus pressure. One study used the International Index of Erectile Function (IIEF) and showed significant improvement (p<0.05).
The studies focused on premature ejaculation noted participants had either lifelong or secondary PE. The pelvic floor training in these studies ranged from 12-20 sessions over 1-3 months. All studies used electrical stimulation as part of the pelvic floor muscle training. Four studies also used biofeedback. Only one study listed a home exercise program but did not report on compliance. The pelvic floor muscle training was compared to nothing in three studies, and to a selective serotonin reuptake inhibitor (SSRI) in the other two studies. Patient reported full resolution of symptoms was 55-83% in two studies, and there was a significant improvement in delay in heterosexual penetrative ejaculation (p<0.05) in three studies.
For both erectile dysfunction and premature ejaculation, pelvic floor muscle exercise prescription was 2-3 times per week with pelvic floor muscle contractions both maximal quick contractions and submaximal endurance holds. Significant results were shown with participants who were taught pelvic floor muscle contractions through a combination of verbal and physical means (typically biofeedback). Specific verbal cues were not reported. The authors suggest that electrical stimulation was helpful for training recruitment patterns; however, there was not a significant difference in outcomes for those with ED when using electrical stimulation. The authors suggest that pelvic floor muscle training can be part of a conservative treatment. It may be used with oral pharmacology for quick results, and may be beneficial with electrical stimulation and biofeedback, though more research is indicated.
If you are interested in learning more about treating male patients, consider attending Male Pelvic Floor: Function, Dysfunction, and Treatment!
Myers, C., Smith, M. “Pelvic floor muscle training improves erectile dysfunction and premature ejaculation: a systematic review” Physiotherapy 105 (2019) 235–243
“However, at present we are all aglow with views on visceral anatomy and medical colleges are wisely establishing chairs in this department which will result in much advancement.” -Fred Byron Robinson, 1891 (Cysts of the Urachus)
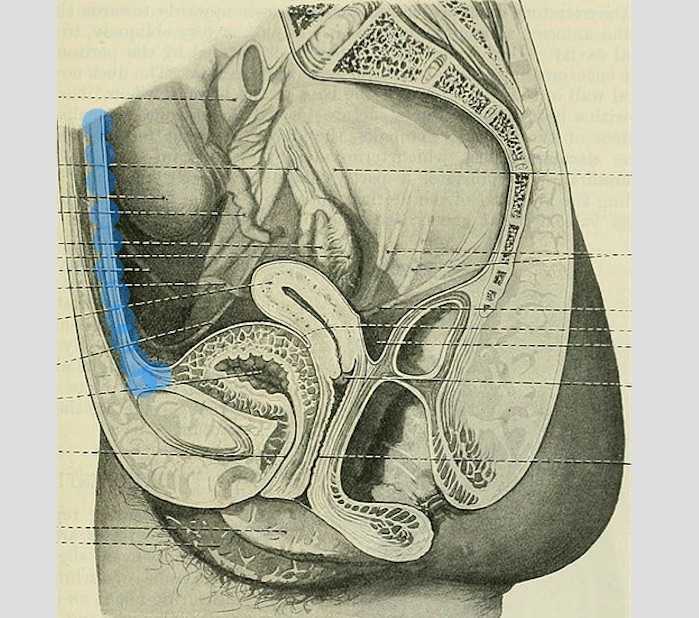
The above (fabulous) quote reminds us that many came before us who were equally excited by the study of anatomy. One anatomical structure that I know never appeared in my graduate anatomy courses is the urachus. The urachus is a structure that extends from the urinary bladder to the umbilicus (highlighted blue in the image). When investigating literature about this structure I was impressed to find publications about the urachus dating from the late 19th century.
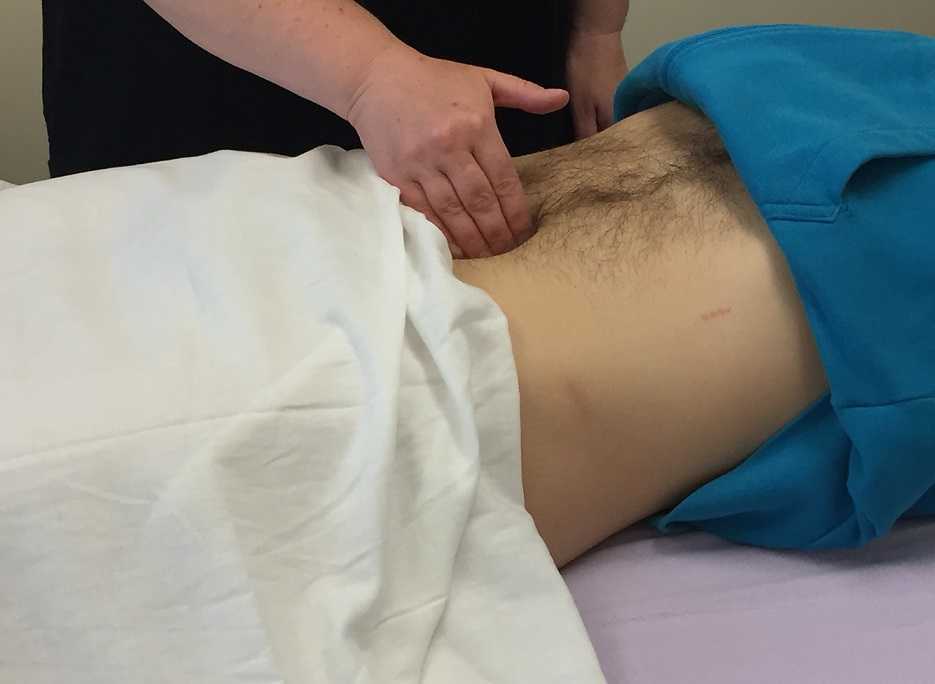
It wasn’t until several years into my career as a physical therapist that I learned about the urachus, a structure attached to the bladder, and learned how this structure could create some rather dramatic symptoms when in dysfunction. I met a woman who was in her early 30’s and who had 6 months prior undergone a laparoscopic surgery with access just below the umbilicus. She presented to rehabilitation after seeing an urologist for severe pain that occurred towards the end of voiding. The pain was absent at any other time, but was so severe towards the latter half of emptying her bladder that she would double over in pain and “nearly pass out.” Investigation revealed a healthy, well-functioning pelvic floor and abdominal wall, but a reproduction of her severe pain with palpation to the midline between the umbilicus and the pubic bone. After grabbing some anatomy texts, I supposed that the urachus, having potentially been irritated by the laparoscopic approach, might experience a tensioning of the irritated tissue as the bladder contracted to empty. This theory appeared to hold some weight, as applying gentle manual therapy to the tissues, and teaching the patient some self-release techniques allowed her to resolve her symptoms entirely after 1-2 visits.
The urachus is formed in early development from the pre-peritoneal layer, and is described as vestigial tissue. It extends from the anterior dome of the bladder to the umbilicus, varying in length from 3-10 cm, with a diameter of 8-10 mm. There are 3 layers: an inner layer of transitional or cuboid epithelial cells surrounded by a layer of connective tissue. The remains of the urachus form the middle umbilical ligament which is a fibromuscular cord. A layer of smooth muscle that is continuous with the detrusor (smooth muscle of the bladder) makes up the outer layer. This continuity of tissue may help explain some of the clinical connections we see in patient symptoms. In the past year, I have met several patients for whom the urachus is the only tissue that reproduced their symptoms. I examined a male patient who reported urethral burning that occurred with both reset and activity. Unable to produce symptoms in any other location of the thoracolumbar spine, pelvic floor and walls, or abdomen, I palpated this structure to find that it reproduced the urethral burning. Another patient presented with a keloid c-section scar. She also described a sharp pain when the bladder was full. Treatment directed to the scar and along the midline resolved this pain, again with a couple sessions.
If you are interested in learning more about distinct anatomical connections that can help you explain (and treat) issues your patients present with, come and learn with us at the 3-day Male Pelvic Floor Function, Dysfunction, and Treatment course offered four times over the 2019 and 2020 calendars. The role of the urachus in abdominopelvic dysfunction is just one of the many topics we explore. With lectures on sexual health, pelvic pain, prostate and urinary dysfunction, there is a broad range of topics and skills to offer for clinicians who are new to men’s heath and those who have been treating for years.
Begg, R. C. (1930). The Urachus: its Anatomy, Histology and Development. Journal of Anatomy, 64(Pt 2), 170–183.
Gray, H. (1918). Anatomy of the Human Body. Philadelphia: Lea & Febiger.
Sterling, J. A., & Goldsmith, R. (1953). Lesions of Urachus which Appear in the Adult. Annals of Surgery, 137(1), 120–128.
At a hair salon, I once saw a plaque that declared, “I’m a beautician, not a magician.” This crossed my mind while reading research on radical prostatectomy, as knowing the baseline penile function of men before surgery seemed challenging. Restoring something that may have been subpar prior to surgery can be a daunting task, and it can cause discrepancies in results of clinical trials. Despite this, two recent studies reviewed the current and future penile rehabilitation approaches post-radical prostatectomy.

Bratu et al.2017 published a review referring to post-radical prostatectomy (RP) erectile dysfunction (ED) as a challenge for patients as well as physicians. They emphasized the use of the International Index of Erectile Function (IIEF) Questionnaire to establish a man’s baseline erectile function, which can be affected by factors such as age, diabetes, alcohol use, smoking habits, heart and kidney diseases, and neurological disorders. The higher the IIEF score preoperatively, the higher the probability of recovering erectile function post-surgery. The experience of the surgeon and the technique used were also factors involved in ED. Radical prostatectomy is a trauma to the pelvis that negatively affects oxygenation of the corpora cavernosum, resulting in apoptosis and fibrotic changes in the tissue, leading to ED. Minimally invasive surgery allows a significantly lower rate of post-RP ED with robot assisted radical prostatectomy (RARP) versus open surgery. The cavernous neurovascular bundles get hypoxic and ischemic regardless of the technique used; therefore, the authors emphasized early post-op penile rehabilitation to prevent fibrosis of smooth muscle and to improve cavernous oxygenation for the potential return of satisfactory sexual function within 12-24 months.
Clavell-Hernandez and Wang2017 [and Bratu et al., (2017)] reported on various aspects of penile rehabilitation after radical prostatectomy. The treatment with the most research to support its efficacy and safety was oral phosphodiesterase type-5 inhibitors (PDE5Is), which help relax smooth muscle and promote erection on a cellular level. Sildenafil, vardenafil, avanafil, and tadalafil have been studied, either used on demand or nightly. Tadalafil had the longer half-life and was considered to have the greatest efficacy. Nightly versus on-demand for any PDE5I was variable in its results. Intracavernosal injection (ICI) and intraurethral therapy using alprostadil for vasodilation improved erectile function, but it caused urethral burning and penile pain. Vacuum erection devices (VED) promoted penile erection via negative pressure around the penis, bringing blood into the corpus cavernosum. There was no need for intact corporal nerve or nitric oxide pathways for proper function, and it allowed for multiple erections in a day. Intracavernous stem cell injections provided a promising approach for ED, and they may be combined with PDE5Is or low-energy shockwave therapy. Ultimately, the authors concluded early penile rehabilitation should involve a combination of available therapies.
Restoring vascularity to healing tissue is a primary goal in rehabilitation, and the sooner the better. Disruption of cavernous nerves and penile tissue post-RP demands rehabilitation, and some methods have more supporting clinical evidence than others. Newer approaches require more exposure and clinical trials for efficacy and long term outcomes. Clinicians should pay attention to updated research and consider taking continuing education courses such as Post-Prostatectomy Patient Rehabilitation or Oncology and the Male Pelvic Floor.
Bratu, O., Oprea, I., Marcu, D., Spinu, D., Niculae, A., Geavlete, B., & Mischianu, D. (2017). Erectile dysfunction post-radical prostatectomy – a challenge for both patient and physician. Journal of Medicine and Life, 10(1), 13–18.
Clavell-Hernández, J., & Wang, R. (2017). The controversy surrounding penile rehabilitation after radical prostatectomy. Translational Andrology and Urology, 6(1), 2–11. http://doi.org/10.21037/tau.2016.08.14