When looking at yoga there are several different types to consider. Some of the most popular are Hatha yoga, Hot yoga, Restorative yoga, Vinyasa yoga, and Yin Yoga. Restorative yoga focuses on relaxing the nervous system and in the 2021 Global Yoga Survey was ranked as the third most popular style worldwide. This survey was given to 10,982 practitioners spanning 124 countries and 29.6% responded that restorative yoga was one of the main styles they practice. (1)
In a dedicated restorative yoga class, a student comes to class, gathers a number of props, and is instructed through 3 to 5 postures, all held for long durations to complete an hour or longer class. Students have various experiences with this type of practice, but over time many begin to feel the need for rest (or restorative practice) in a similar way that one feels thirsty or hungry.
Restorative Yoga and Physical Therapy
The U.S. military, the National Institutes of Health, and other large organizations are listening to, and incorporating, scientific validation of yoga’s value in health care. Numerous studies show yoga’s benefits in arthritis, osteopenia, balance issues, oncology, women’s health, chronic pain, and other specialties. (2)
Restorative yoga can be a valuable adjunct for rehab practitioners in helping patients improve their physical and mental health. It can be especially helpful for people recovering from injuries, surgery, or chronic conditions. Restorative yoga can also be used for active recovery. This practice enables them to be lightly active while allowing their body to heal from strenuous training sessions. Runners, bodybuilders, and other types of athletes can benefit from using restorative yoga for this purpose.
Restorative yoga allows us to access the ventral vagal aspect of the parasympathetic nervous system. This pathway is necessary for healing, stress management, and the ability to choose our actions rather than react. Restorative yoga is an accessible method that may be a new tool in a patient’s toolbox for managing their nervous systems.
The goals of both therapy and restorative yoga are similar in healing and alleviating stress on the body. Remember that restorative yoga teaches people to be more aware of their body and the connection between their body and mind. By combining the two, patients can move forward in their lives and learn how to cope and be aware of what is going on with their bodies.
By adding restorative yoga to your toolkit, you can help your patients learn more about their bodies and pain. It can improve their focus, stability, flexibility, and strength – all of which will improve their results and quality of life.
Join Kate Bailey to learn more about restorative yoga and how to incorporate it into your practice in her upcoming course Restorative Yoga for Physical Therapists (and yes this course is open to all licensed practitioners) on March 22, 2025.
References:
- The global yoga survey 2021: How and why people practice yoga. DoYou. (2022, April 12). Retrieved January 17, 2025, from https://www.doyou.com/yoga-survey/
- 9 Benefits of Yoga. John Hopkins Medicine. Retrieved January 17, 2025 from https://www.hopkinsmedicine.org/health/wellness-and-prevention/9-benefits-of-yoga
AUTHOR BIO:
Kate Bailey, PT, DPT, MS, E-RYT 500, YACEP, Y4C, CPI


I’ve been “on the record saying” I used to be a modalities skeptic. I didn’t think I used them a ton in my practice and when I switched from a big hospital system to my own private practice, I didn’t have access to the larger equipment machines like the big ultrasound or biofeedback devices.
I have, however, always been a fan of gadgets and tools as a pelvic health provider. For anyone who has ever been to a class where I was a teaching assistant, you’ve likely seen me literally roll in with a giant suitcase full of example things - models, pelvic trainers, examples of belts, braces, dilators, wands, weights, lube samples, pelvic organ stuffies etc. I always called it my toolkit and so many of my peers had this as well. When the Content & Curriculum team asked me to be on the team for writing for this project, I was initially unsure. I didn’t think I used modalities enough or valued them enough to create a two-day course on the topic.
Long story short…I was wrong. What I have learned is I was very wrong, and I use them constantly.
As we created the outline for Modalities and Pelvic Function, it became very clear that I had not really understood the definition of a modality. Spoiler alert, we spend the first lecture talking all about this. Once I understood that a modality is basically anything we use to create a change with a patient, I realized I LOVED them and used them all day, every day. I happily settled into writing with the team, reaching out to companies for samples, trial devices, and helped to make this AMAZING toolkit for a class that is like the biggest game of pelvic health show and tell.
I was lucky enough to be one of the instructors for the inaugural class and have been present at every class since. What I did not expect from this class was the “aha” moments and how well these helped students put it all together. Feedback-wise, our team always gets requests to “show more treatment” and “give skills to bring back to the clinic for treatment.” Welcome to two straight days of this for every pelvic topic we could think of in reference to bowel, bladder, and sexual dysfunction while also making room for oddball topics like patient education, models, and handouts.
If you have questions about this course, then look through the following FAQs. If you don’t see your question answered, then reach out to the Herman & Wallace team and they can answer it for you!
Will this class make me buy something?
Nope, no purchase is necessary, but we did work with a whole bunch of companies to get you some free samples or really good discounts if you want your own.
Does my pelvic floor have to be available for this class?
We do two labs with internal sensors so a provider can feel what biofeedback and e-stim feel like. You get to choose your sensor and preferred canal and apply your own device. For those who are a little shy about sharing their pelvis for the sake of science, I’ve been told this has been a really safe feeling option. If you’re not up for an insertable device, we offer external options as well.
What skills will I take home?
You’ll learn all the features of every modality that our writing team could think of all organized by topic and primary treatment usage (don’t worry, we’ll also share our pearls and hacks.) We’ll also talk about ways to teach your patients about these devices including why a device might be indicated, how it can be used, cleaned, etc.
Is this class for newbies or those who are seasoned?
Yes! We’ve had participants of all different experiences and skill levels find this class extremely useful and a great resource for their practice. If you work in pelvic health and are an expert in gadgets already, this may NOT be the course for you unless you’d like to be one of our awesome teaching assistants.
We’ve worked so hard to make Modalities and Pelvic Function a well-rounded learning experience. This class is ONLY in person and tends to be a very intimate group of providers, which was another request from our Herman & Wallace customers. We hope to see you at one of the five offerings of Modalities and Pelvic Function this year!
- Galloway, NJ - February 8-9
- Sacramento, CA - April 5-6
- Houston, TX - July 12-13
- Akron, OH - September 20-12
- Somerset, NJ - October 4-5
AUTHOR BIO
Mora Pluchino, PT, DPT, PRPC
 Mora Pluchino, PT, DPT, PRPC (she/her) is a graduate of Stockton University with a BS in Biology (2007) and a Doctorate of Physical Therapy (2009). She has experience in a variety of areas and settings, working with children and adults, including orthopedics, bracing, neuromuscular issues, vestibular issues, and robotics training. She began treating Pelvic Health patients in 2016 and now has experience treating women, men, and children with a variety of Pelvic Health dysfunction. There is not much she has not treated since beginning this journey and she is always happy to further her education to better help her patients meet their goals.
Mora Pluchino, PT, DPT, PRPC (she/her) is a graduate of Stockton University with a BS in Biology (2007) and a Doctorate of Physical Therapy (2009). She has experience in a variety of areas and settings, working with children and adults, including orthopedics, bracing, neuromuscular issues, vestibular issues, and robotics training. She began treating Pelvic Health patients in 2016 and now has experience treating women, men, and children with a variety of Pelvic Health dysfunction. There is not much she has not treated since beginning this journey and she is always happy to further her education to better help her patients meet their goals.
She strives to help all of her patients return to a quality of life and activity that they are happy with for the best bladder, bowel, and sexual functioning they are capable of at the present time. In 2020, She opened her own practice called Practically Perfect Physical Therapy Consulting to help meet the needs of more clients. She has been a guest lecturer for Rutgers University Blackwood Campus and Stockton University for their Pediatric and Pelvic Floor modules since 2016. She has also been a TA with Herman & Wallace since 2020 and has over 150 hours of lab instruction experience. Mora has also authored and instructs several courses for the Institute.

Kathleen D. Gibson, MD, Rachna Mehta PT, DPT, CIMT, OCS, PRPC, RYT 200, and Mora Pluchino, PT, DPT, PRPC are joining our esteemed lineup of speakers and session leaders for HWConnect 2025 scheduled for March 28-30, 2025. Their expertise and unique approaches to healthcare will provide invaluable insights for all attendees.
Kathleen D. Gibson, MD: Pelvic Venous Disease (PeVD) and Chronic Pelvic Pain
 Dr. Gibson will be presenting "Pelvic Venous Disease (PeVD) and Chronic Pelvic Pain: A Multidisciplinary Approach to Diagnosis and Treatment." This topic is critical for understanding the complexities of pelvic venous disease, a common yet often underdiagnosed condition that can cause chronic pelvic pain. Dr. Gibson’s presentation will delve into the latest diagnostic methods and treatment options available for PeVD, highlighting the importance of a multidisciplinary approach for optimal patient care. Whether you're a clinician, researcher, or patient advocate, this session will provide practical insights that can make a real difference in your practice.
Dr. Gibson will be presenting "Pelvic Venous Disease (PeVD) and Chronic Pelvic Pain: A Multidisciplinary Approach to Diagnosis and Treatment." This topic is critical for understanding the complexities of pelvic venous disease, a common yet often underdiagnosed condition that can cause chronic pelvic pain. Dr. Gibson’s presentation will delve into the latest diagnostic methods and treatment options available for PeVD, highlighting the importance of a multidisciplinary approach for optimal patient care. Whether you're a clinician, researcher, or patient advocate, this session will provide practical insights that can make a real difference in your practice.
Rachna Mehta PT, DPT, CIMT, OCS, PRPC, RYT 200: Acupressure Self-Regulation Acupoints
 Dr. Mehta, a highly experienced physical therapist and integrative medicine practitioner, will lead a breakout session on Acupressure Self-Regulation Acupoints. This hands-on session will introduce participants to powerful acupressure techniques designed to promote healing, reduce stress, and enhance overall well-being. Attendees will learn how to use acupressure for self-regulation, a simple and effective tool to manage pain, anxiety, and other health concerns. Dr. Mehta’s expertise in integrating physical therapy with traditional healing practices makes this a session you won’t want to miss.
Dr. Mehta, a highly experienced physical therapist and integrative medicine practitioner, will lead a breakout session on Acupressure Self-Regulation Acupoints. This hands-on session will introduce participants to powerful acupressure techniques designed to promote healing, reduce stress, and enhance overall well-being. Attendees will learn how to use acupressure for self-regulation, a simple and effective tool to manage pain, anxiety, and other health concerns. Dr. Mehta’s expertise in integrating physical therapy with traditional healing practices makes this a session you won’t want to miss.
Mora Pluchino, PT, DPT, PRPC: The Pelvic Rehab Provider’s Role in Infant Care
 Dr. Pluchino is a leading expert in pelvic rehabilitation, will be presenting "The Pelvic Rehab Provider’s Role in Infant Care: Managing Reflux, Constipation, and “Colicky” Babies." This session is a must-attend for providers looking to expand their practice into the world of infant care and address some of the most common gastrointestinal (GI) challenges affecting newborns and their families. GI distress in infants is a common but often overwhelming challenge for caregivers. As pelvic rehab providers, we’re uniquely positioned to make a meaningful difference in the lives of these families. By learning targeted strategies for infant care, you can expand your skillset, offer more comprehensive services, and positively impact your youngest patients. This session will feature hands-on demonstrations, including abdominal massage for Infants, positioning strategies, as well as play and movement techniques.
Dr. Pluchino is a leading expert in pelvic rehabilitation, will be presenting "The Pelvic Rehab Provider’s Role in Infant Care: Managing Reflux, Constipation, and “Colicky” Babies." This session is a must-attend for providers looking to expand their practice into the world of infant care and address some of the most common gastrointestinal (GI) challenges affecting newborns and their families. GI distress in infants is a common but often overwhelming challenge for caregivers. As pelvic rehab providers, we’re uniquely positioned to make a meaningful difference in the lives of these families. By learning targeted strategies for infant care, you can expand your skillset, offer more comprehensive services, and positively impact your youngest patients. This session will feature hands-on demonstrations, including abdominal massage for Infants, positioning strategies, as well as play and movement techniques.
The combination of Dr. Gibson's groundbreaking work in pelvic health, Dr. Mehta's holistic approach to self-care, and Dr. Pluchino’s pediatric session on infant care adds a well-rounded, comprehensive experience for anyone interested in improving patient outcomes and understanding new ways to support holistic health. This is a unique opportunity to hear from three thought leaders who bring fresh perspectives to modern healthcare.
Join us at HWConnect 2025 on March 28-30 in Seattle WA to connect, learn, and grow with leading experts in the field! For more information and to register for HWConnect 2025, visit https://www.hermanwallace.com/hwconnect-2025. We can’t wait to see you there!

A pessary is a device that is placed in the vaginal canal to support the pelvic organs. They can be worn only during an activity that typically provokes symptoms, such as running, or used almost continuously with periodic cleaning. People looking to avoid or delay pelvic organ prolapse surgery, such as those planning to give birth, may especially benefit from pessary use. Many transgender and nonbinary people with a front canal also find pessary use helpful for reducing POP symptoms.
Pessaries have additional benefits beyond improving prolapse symptoms. For example, some pessaries are designed to mitigate urinary incontinence by applying pressure to the urethra (1). A 2024 study even indicated that they may improve sexual wellness in people with pelvic organ prolapse (2). One case study series suggests that intersex people who have undergone vaginoplasty or neovaginoplasty may also benefit from pessary use for maintaining the patency of the canal (5).
Traditionally in the United States, pessaries have been placed by medical professionals rather than rehabilitation professionals (although some PTs at the Veterans Affairs were involved in fitting pessaries under the guidance of urogynecologists). In 2021, the American Physical Therapy Association’s Academy of Pelvic Health convened a Pessary Task Force to look at the feasibility of physical therapists fitting and managing pessaries. In 2022, the Academy of Pelvic Health released its position statement to include pessary fitting and management in the scope of practice of pelvic health physical therapists in the United States and its five territories. By the end of 2022, the Academy of Pelvic Health hosted its first pessary fitting course. Several more courses have been offered since then. Physical therapists specialize in showing clients how to tend to and support their bodies. Active participation in pessary fitting and management is a way to help the nearly 40% of people with vaginal canals who are expected to develop pelvic organ prolapse.
If you would like to learn more, Herman & Wallace now has a pessary course! Pessaries and Pelvic Rehab is a two-day in-person course designed for pelvic therapists interested in adding pessary fitting to their clinical practice and have completed Pelvic Function Level 1 and Level 2B. Pessaries and Pelvic Rehab is scheduled for Chicago, IL on November 8-9, 2025. We look forward to seeing you there!
Note: HW does not currently know of any occupational therapists fitting pessaries in the United States, but they hope that this becomes a part of OT scope in the future.
References
- Shah SM, Sultan AH, Thakar R. The history and evolution of pessaries for pelvic organ prolapse. Int Urogynecol J Pelvic Floor Dysfunct. 2006;17(2):170-175.
- Jin C, Yan H, Shang Y, et al. Real-world clinical effectiveness of nonsurgical treatments for female with POP-Q stage II cystocele: a retrospective analysis of therapeutic efficacy. Transl Androl Urol. 2024;13(4):483-492.
- Petter Rodrigues M, Bérubé MÈ, Charette M, McLean L. Conservative interventions for female exercise-induced urinary incontinence: a systematic review. BJU Int. Published online July 23, 2024.
- Nemeth Z, Vida P, Markovic P, Gubas P, Kovacs K, Farkas B. Long-term self-management of vaginal cube pessaries can improve sexual life in patients with pelvic organ prolapse, results from a secondary analysis. Int Urogynecol J. Published online August 5, 2024.
- Mensah V, Christianson MS, Yates M, Tobler K, Kolp LA. Novel use of a pessary to maintain vaginal patency following vaginoplasty or neovaginoplasty for mullerian anomalies or agenesis. Fertility and Sterility. 2013;99(3):S37.
- Wang B, Chen Y, Zhu X, et al. Global burden and trends of pelvic organ prolapse associated with aging women: An observational trend study from 1990 to 2019. Front Public Health. 2022;10:975829.

Before we talk about yoga, let’s do a quick overview of interstitial cystitis/bladder pain syndrome.
In 2022, the American Urological Association updated the clinical guidelines on the treatment of interstitial cystitis/bladder pain syndrome. Prior to this revision, pelvic physical (and occupational) therapy was considered first-line treatment with evidence strength grade A. After this revision, the AUA recommends looking at the phenotype of the patient to determine which treatment categories would best serve the individual patient.
Three phenotypes have been identified: a bladder-centric phenotype, a pelvic floor phenotype, and a phenotype that experiences widespread symptoms with chronic overlapping pain conditions.
Treatment categories include behavioral/non-pharmacologic, oral medicines, bladder instillations, procedures, and major surgery.
There are three guideline statements that are within our scope of practice as rehab professionals.
Guideline Statement 10: “Self-care practices and behavioral modifications that can improve symptoms should be discussed and implemented as feasible. Clinical Principle”
This is the space that pelvic PTs and OTs live in every day. The recommended treatments within our scope of practice include:
- evaluating the concentration and/or volume of urine
- limiting bladder irritants
- an elimination diet to determine which foods or fluids are triggering
- thermal modalities (eg, suprapubic or perineal heat or cold)
- strategies to manage IC/BPS flare-ups (eg, meditation, imagery)
- pelvic floor muscle relaxation
- bladder retraining
- avoiding pain-provoking exercise
- managing constipation
Guideline Statement 12: “Appropriate manual physical therapy techniques (eg, maneuvers that resolve pelvic, abdominal, and/or hip muscular trigger points, lengthen muscle contractures, and release painful scars and other connective tissue restrictions), if appropriately trained clinicians are available, should be offered to patients who present with pelvic floor tenderness. Pelvic floor strengthening exercises (eg, Kegel exercises) should be avoided. Standard (Evidence Strength: Grade A)”
As therapists, we will often also evaluate the spine and perhaps further up and down the chain to look for potential contributing factors. While this was not mentioned in the guideline paper, it is often how we holistically treat each individual who walks into our office.
But wait! There’s more! We can also help coach our patients on one more clinical principle….
Guideline Statement 11: “Patients should be encouraged to implement stress management practices to improve coping techniques and manage stress-induced symptom exacerbations. Clinical Principle”
Remember back in guideline 10 when it listed “strategies to manage IC/BPS flare-ups (eg, meditation, imagery) and pelvic floor muscle relaxation”? Clinically we use breathwork, pain neuroscience, and gentle stretching. Some practitioners use yoga as an adjunct modality in the clinic and for home programs.
Khandwala et al looked at 8 patients during 3 months of integrated yoga therapy. There was a statistically significant improvement in two areas of the SF-36 (social function and pain). There was a non-statistically significant trend towards improved voiding volumes.
Dao et al looked at 97 patients assigned to the control group (usual care) or intervention group (usual care plus yoga and meditation). The intervention group showed faster improvement during the clinical trial. Additionally, there were decreases in anxiety, depression, pain interference, and pain scores.
I (Dustienne) created the two flows for the Dao study, and I’m absolutely thrilled to see echoed in the research what we see clinically - downregulation of the nervous system, breathwork, visualization, and mindful movement support our patients and offer additional strategies for resilience.
For free access to the two videos used in the study, visit: https://www.yourpaceyoga.com/interstitial-cystitis-yoga
Dustienne Miller developed her course Yoga For Pelvic Pain in 2012. She enjoys seeing the research grow and supports how she and her colleagues have been practicing in pelvic health over the years. Remote course offerings for 2025 are: February 22-23, May 3-4, and September 13-14.
References:
- Clemens, J. Q., Erickson, D. R., Varela, N. P., & Lai, H. H. (2022). Diagnosis and Treatment of Interstitial Cystitis/Bladder Pain Syndrome. Journal of Urology, 208(1), 34–42. https://doi.org/10.1097/JU.0000000000002756 (Original work published July 1, 2022)
- Dau, A., Meriwether, K.V., Petersen, T., Jansen, S. & Komesu, Y. Mindfulness and Yoga for Pain with Interstitial Cystitis Evaluation: An RCT
- FitzGerald, M. P., Anderson, R. U., Potts, J., Payne, C. K., Peters, K. M., Clemens, J. Q., ... & Nyberg, L. M. (2009). Urological Pelvic Pain Collaborative Research Network Randomized multicenter feasibility trial of myofascial physical therapy for the treatment of urological chronic pelvic pain syndromes. J. urol, 182(2), 570-580.
- Khandwala, S., & Cruff, J. (2020). The Role of Yoga in the Management of Bladder Pain Syndrome: A Single-Arm Pilot Study. Advances in mind-body medicine, 34(4), 4–9.
- Rovner, E., Propert, K. J., Brensinger, C., Wein, A. J., Foy, M., Kirkemo, A., ... & Interstitial Cystitis Data Base Study Group. (2000). Treatments used in women with interstitial cystitis: the interstitial cystitis database (ICDB) study experience. Urology, 56(6), 940-945.
Author Bio:
Dustienne Miller PT, MS, WCS, CYT

Dustienne Miller is the creator of the two-day course Yoga for Pelvic Pain and an instructor for Pelvic Function Level 1. Born out of an interest in creating yoga home programs for her patients, she developed a pelvic health yoga video series called Your Pace Yoga in 2012. She is a contributing author in two books about the integration of pelvic health and yoga, Yoga Mama: The Practitioner’s Guide to Prenatal Yoga (Shambhala Publications, 2016) and Healing in Urology (World Scientific). Prior conference and workshop engagements include APTA's CSM, International Pelvic Pain Society, Woman on Fire, Wound Ostomy and Continence Society, and the American Academy of Physical Medicine and Rehabilitation Annual Assembly.
Her clinical practice, Flourish Physical Therapy, is located in Boston's Back Bay. She is a board-certified women's health clinical specialist recognized by the American Board of Physical Therapy Specialties. Dustienne weaves yoga, mindfulness, and breathwork into her clinical practice, having received her yoga teacher certification through the Kripalu Center for Yoga and Health in 2005.
Dustienne's love of movement carried over into her physical therapy and yoga practice, stemming from her previous career as a professional dancer. She danced professionally in New York City for several years, most notably with the national tour of Fosse. She bridged her dance and physical therapy backgrounds working for Physioarts, who contracted her to work backstage at various Broadway shows and for Radio City Christmas Spectacular. She is an assistant professor of musical theater and jazz dance at the Boston Conservatory at Berklee.
Dustienne passionately believes in the integration of physical therapy and yoga within a holistic model of care. Her course aims to provide therapists and patients with an additional resource centered on supporting the nervous system and enhancing patient self-efficacy.

When we consider the intersectionality of sexual orientation and gender identity within the realm of intersex and LGBTQIA+ identities, we must recognize the multifaceted nature of individuals' experiences. For some intersex individuals, navigating aspects of identity may be complex, as their biological sex characteristics may not align with societal expectations of gender. Intersex individuals face a range of challenges in navigating their identities, including medical interventions without their consent, stigmatization, and lack of awareness and understanding from healthcare providers. These challenges highlight the need for clinicians to educate themselves on intersex experiences and provide inclusive and affirming care.
Resources for Clinicians to Support Intersex Patients
 While you may be reading this thinking, ‘I don’t know anyone who is intersex,’ or ‘I don’t work with Intersex patients,’ you might be surprised to learn that you probably do! Intersex variations are as common as having red hair. The United Nations Free & Equal Initiative Intersex Fact Sheet states that “According to experts, somewhere between 0.05 percent and 1.7 percent of the global population is born with intersex traits. The upper estimate is bigger than the population of Mexico.” So, if you know, or have ever seen someone with red hair, you probably know or have seen someone who is intersex too.
While you may be reading this thinking, ‘I don’t know anyone who is intersex,’ or ‘I don’t work with Intersex patients,’ you might be surprised to learn that you probably do! Intersex variations are as common as having red hair. The United Nations Free & Equal Initiative Intersex Fact Sheet states that “According to experts, somewhere between 0.05 percent and 1.7 percent of the global population is born with intersex traits. The upper estimate is bigger than the population of Mexico.” So, if you know, or have ever seen someone with red hair, you probably know or have seen someone who is intersex too.
Clinicians who are looking to better support their intersex patients can benefit from accessing resources specifically tailored to understanding and addressing the unique needs of this community. Two great Intersex-led organizations to help educate you and provide support to intersex patients and their caregivers are InterACT Advocates for Intersex Youth and InterConnect.
These organizations offer information and guidance on how to provide affirming care to intersex individuals and provide help to clinicians who want to educate themselves on intersex identities, familiarize themselves with the challenges faced by intersex individuals, and learn about best practices for promoting the health and well-being of their intersex patients.
Educate yourself, stand with intersex advocacy organizations, and amplify intersex voices to make a difference. Let's strive for a world where every identity is celebrated and respected because diversity strengthens us all. As Maya Angelou once said, "We all should know that diversity makes for a rich tapestry, and we must understand that all the threads of the tapestry are equal in value no matter their color."
So how do I learn more? What resources can I give to intersex patients and their families to support them? Well, to learn more about how to be an ally in healthcare to intersex patients and their families, and for more resources, register today for Intersex Patients: Rehab and Inclusive Care, scheduled for February 8th, 2025!
Additional Resources:
- InterACT Advocates, Lambda Legal (2018). Providing Ethical and Compassionate Health Care to Intersex Patients: Intersex-Affirming Hospital Policies. This guide incorporates input from medical practitioners, legal experts, and members of the intersex community. The guide offers model policies for hospitals designed to promote best practices and assist hospitals and their providers in delivering appropriate, intersex-affirming care. For example, the policies address issues of confidentiality, non-discrimination, gender identification, infant genital surgery, and sterilization, shared decision-making, and informed consent – issues that, when mishandled, can cause significant harm to intersex patients and their families, as well as open up medical institutions to significant liability. The guide also incorporates background information and explanations for each model policy to provide education in an area that has historically been misunderstood.
- Intersex Peer Support Australia (IPSA). https://isupport.org.au/. IPSA is a non-profit organization, led by people with variations of sex characteristics (sometimes known as intersex) who are passionate about combatting isolation, shame, and stigma through community connection and peer support. IPSA seeks to tackle the stigma and misconceptions that surround intersex variations through education and advocates on issues affecting the wider intersex community to improve affirmative healthcare, foster intersex pride, strengthen our community, and deepen social culture. IPSA is a peer-led, not-for-profit intersex organization and a registered health promotion charity.
- InterLink. https://www.ilink.net.au/. InterLink brings people together to talk about living with innate variations in sex characteristics with the support of trained counselors and intersex peer workers. InterLink also provides community care coordination, helping people get linked in with appropriate allied health and community-based services, advocacy, and peer support groups.

One of the most bothersome and common symptoms experienced by patients going through the menopausal transition is hot flashes (Freedman 2015). Vasomotor symptoms can vary in intensity from mild to debilitating (Gold et al., 2000), and patients can suffer from a flushed face up to a full sweat with the removal of clothing and a brisk fan required for relief. Hot flashes can affect a patient’s focus, sleep, and activity tolerance. These pesky flashes are also associated with several medical disorders including heart disease, dementia, and osteoporosis (Biglia et al, 2017).
What causes hot flashes isn’t entirely known. A variety of factors can be at play including genetics, personal experience, cultural influences, and medications (Biglia et.al, 2017), however, one of the predominant factors contributing to these flushes is decreasing or fluctuating estrogen levels. Declining estrogen is linked with the KnDy (kisspeptin-neurokinin B-dynorphin neurons) located in the hypothalamus. These neurons project to the thermoneutral zone also located in the hypothalamus. This zone regulates the temperature in the body. As estrogen levels diminish, these neurons hypertrophy. This causes an increase in activity to the thermoneutral zone making the patient more sensitive to temperature changes (Rance et al, 2013). A small shift in temperature causes a greater physiological response triggering the hot flash.
In their 2022 position statement on hormone therapy, the North American Menopause Society recommends estrogen as one of the most effective treatments for this symptom. It is cited as a safe and effective option which many choose for relief. For some patients, this is not an option due to either personal choice or contraindications from their medical history.
An adjunct or alternative treatment for hot flashes is cognitive behavioral therapy (CBT). It has been proven as an effective diminisher of hot flashes and can be utilized by patients through this transition (The Non-Hormonal Position Statement of the North American Menopause Society 2023). In the book Living Well Through the Menopause, authors Hunter and Smith describe the importance of utilizing cognitive behavioral therapy as a tool for diminishing the intensity and bother of hot flashes.
One of the tools specifically mentioned in their recommendations is diaphragmatic breathing. This is a common skill that can be taught to patients by providers to help manage pain and urinary urgency symptoms. By tapping into the parasympathetic or “rest and digest” aspect of the autonomic nervous system, it facilitates the body to chill out and calm. This can also be recommended to patients in the menopausal transition as one method of hot flash management. Quieting the nervous system throughout the day can aid in stress management and decrease the intensity of hot flashes.
Another tool for management is self-care (Hunter and Smith 2021). Perimenopause can be a time of great stress for many. Busy work schedules, aging parents, and active teenagers can cause patients to forget about prioritizing time for themselves to reflect, recharge, and pause. Patients' lives are often constant caregiving and chaos. With this flurry of activity, the importance of their own health and well-being can be forgotten. Clinicians are integral in reminding patients that taking time for themselves will ensure they are capable of handling the circus of activities they are juggling. Giving the patient permission for self-care can be invaluable. Encouraging exercise, friendships, and taking a rest can help with stress management and in turn can help with sleep and the severity of symptoms (Hunter and Smith 2021).
Ahhhhh sleep, so often disrupted in this phase of life. Hot flashes certainly play a role with this as does stress. Both can play off the other. Educating our patients about the effects of alcohol, caffeine, and bright light bombardment before bed can help them on the road to better rest. Teaching meditation or diaphragmatic breathing before bed can also provide benefits (Hunter and Smith 2021). With better sleep comes less stress and with less stress comes reduced symptoms.
When patients experience vasomotor symptoms, the pelvic health provider has several tools in their toolbox to help with management. There are non-hormonal options out there that can make a difference. Clinicians can help patients navigate through the menopausal transition with tools for decreasing the intensity of symptoms and improving quality of life.
To learn more, sign up for Menopause Transitions and Pelvic Rehab scheduled for February 1-2, 2025. This course is an excellent opportunity to understand the physiological consequences to the body as hormones decline, in order to assist our patients in lifestyle habits for successful aging. Course topics include cardiovascular changes, metabolic syndrome, bone loss and sarcopenia, neurological changes (headache, brain fog, sleeplessness), Alzheimer’s risk, and urogenital changes. Symptoms and treatment options will also be discussed, including hormone replacement, non-hormonal options, dietary choices, and exercise considerations.
References:
- The 2022 hormone therapy position statement of The North American Menopause Society. Menopause, 2022. 29(7): p. 767-794.
- Biglia, N., et al., Vasomotor symptoms in menopause: a biomarker of cardiovascular disease risk and other chronic diseases? Climacteric, 2017. 20(4): p. 306-312.
- Freedman, R.R., Menopausal hot flashes: mechanisms, endocrinology, treatment. J Steroid Biochem Mol Biol, 2014. 142: p. 115-20.
- Gold, E.B., et al., Relation of demographic and lifestyle factors to symptoms in a multi-racial/ethnic population of women 40-55 years of age. Am J Epidemiol, 2000. 152(5): p. 463-73.
- Hunter, M.a.S., Melanie, Living Well Through the Menopause: An Evidenced Based Cognitive Behavioural Guide 2021, Great Britain: Robinson.
- Rance, N.E., et al., Modulation of body temperature and LH secretion by hypothalamic KNDy (kisspeptin, neurokinin B and dynorphin) neurons: a novel hypothesis on the mechanism of hot flushes. Front Neuroendocrinol, 2013. 34(3): p. 211-27.
- The Nonhormone Therapy Position Statement of The North American Menopause Society" Advisory, P., The 2023 nonhormone therapy position statement of The North American Menopause Society. Menopause, 2023. 30(6): p. 573-590.
Author Bio
Christine Stewart, PT, CMPT
 Christine Stewart, PT, CMPT (she/her) graduated from Kansas State University in 1992 and went on to pursue her master’s degree in physical therapy from the University of Kansas Medical Center graduating in 1994. She began her career specializing in orthopedics and manual therapy then became interested in women’s health after the birth of her second child.
Christine Stewart, PT, CMPT (she/her) graduated from Kansas State University in 1992 and went on to pursue her master’s degree in physical therapy from the University of Kansas Medical Center graduating in 1994. She began her career specializing in orthopedics and manual therapy then became interested in women’s health after the birth of her second child.
Christine developed her pelvic health practice in a local hospital with a focus on urinary incontinence and prolapse. She left the practice in 2010 to work at Olathe Health to further focus on pelvic rehabilitation for all genders and obtain her CMPT from the North American Institute of Manual Therapy. She completed Diane Lee’s Integrated Systems Model education series in 2018. Her passion is empowering patients through education and treatment options for the betterment of their health throughout their lifespan. She enjoys speaking to physicians and to community-based organizations on pelvic health physical therapy.

Did you know that Ramona Horton is going to be speaking at HWConnect in March? Her lecture is titled, “The Do Not Miss List: What many pelvic rehab therapists overlook." I don’t know about you, but we’re pretty excited to hear what she has to say and to learn from the best!
So, who is Ramona C. Horton MPT, DPT?
Ramona completed her graduate training in the US Army–Baylor University Program in Physical Therapy in San Antonio, TX. She exited the army at the rank of Captain and applied her experience with the military orthopedic population in the civilian sector as she developed a growing interest in the field of pelvic dysfunction and received her post-professional Doctorate in Physical Therapy from A.T. Still University in Mesa, AZ. In 2020, Ramona received the prestigious Academy of Pelvic Health Elizabeth Noble Award for her contributions to the field of pelvic health.
Ramona is the lead therapist for her clinic's pelvic dysfunction program in Medford, OR where her practice focuses on the treatment of urological, gynecological, and colorectal issues. Ramona has completed advanced studies in manual therapy with an emphasis on spinal manipulation, and visceral and fascial mobilization.
Not only is Ramona Horton, MPT, DPT speaking at HWConnect 2025 in March, but she has also developed and instructs the Visceral and Fascial Mobilization Course Series for Herman & Wallace. If you haven’t taken a course from Ramona or heard her speak, then we highly recommend that you do!
The top 3 reasons to sign up for a course with Ramona Horton are:
1. Understand the true function and mechanisms of manual therapy.
Manual therapy is presented as a concept and technique that does NOT “release” tight or bound fascia based on the skill or magic hands of the practitioner. The issue is not in the tissue, if the tissue is tight, it’s tight because the brain is keeping it that way. Muscles are marionettes, and the brain is the puppet master. Manual therapy utilizes the fascial system to access the nervous system. In other words, having a conversation with the brain over the tissue that it appears to be protecting while trusting that the homeostatic mechanism is functioning in the body. If this is done in a non-threatening manner, the brain will normalize the tissue it is holding and guarding.
2. Add a whole host of new tools to your practitioner toolbelt.
The myofascial course teaches basic screening techniques that will point you in the right direction toward finding where the body is protecting, not where symptoms are being expressed. You will learn a variety of techniques to approach different fascial layers including direct and indirect fascial stacking for superficial nerves within the panniculus, muscular, and articular restrictions, as well as indirect technique of positional inhibition for trigger points. In addition, the science behind basic neural mobilization, instrument-assisted fascial mobilization, and fascial decompression (cupping) are presented.
3. Learn more about fascia, its origins, and its functions.
Fascia is EVERYWHERE throughout the body; it is the ubiquitous connective tissue that holds every cell together much like the mortar in a brick wall, in addition to cells, it connects every system in the body. Fascia contains a vast neurological network including nociceptors, mechanoreceptors, and proprioceptors just to name a few. The fascial system has multiple layers within the body: starting at the panniculus which blends with the skin, the investing fascia surrounding muscles and forming septae, the visceral fascia which is by far the most complex and the deepest layer of fascia, the dura surrounding the central nervous system extending to the peripheral nerves. All fascial structures, regardless of layer or location have their origin in the mesoderm of early embryologic development. The myofascial course presents evaluation and treatment techniques for three of the four fascial layers while the three visceral courses address the complex visceral fascial layer.
Ramona Horton's Mobilization Series 2024 Course Schedule
The Mobilization courses are available in satellite and self-hosted formats. PLUS Ramona is going on the road this year and will be teaching directly from different satellites for each course. Find out more on the Visceral and Fascial Mobilization Course Series home page. Satellite locations can be found on the main course page and may change, be added or removed, for future course events.
Mobilization of the Myofascial Layer: Pelvis and Lower Extremity - Satellite Lab Course
April 4-6
Bradenton FL
Medford OR
Milwaukee WI
Novato CA
St. Petersburg FL
Torrance CA
Self-Hosted
Mobilization of Visceral Fascia: The Gastrointestinal System - Satellite Lab Course
March 7-9
Appleton WI
Lansing MI
Nashville TN
Portland ME
Tampa FL
Torrance CA
Tuscon AZ
Self-Hosted
June 27-29
Milwaukee WI
St. Petersburg FL
Sellersville PA
Self-Hosted
Mobilization of Visceral Fascia: The Urinary System - Satellite Lab Course
January 31-February 2
Fort Lauderdale FL
Medford OR
Tampa FL
Torrance CA
Wichita KS
Self-Hosted
May 16-18
Atlanta GA
Bradenton FL
Philadelphia PA
Self-Hosted
November 14-16
Milwaukee WI
Stevens Point WI
Self-Hosted
Mobilization of Visceral Fascia: The Reproductive System - Satellite Lab Course
October 17-19
Milwaukee WI
Omaha NE
Torrance CA
Tuscon AZ
Self-Hosted

Mental Health is a very serious global concern! As a health care provider, I have seen my patients anxious, scared, confused, tearful, depressed, and sometimes in sheer panic with debilitating anxiety and pain. As they express these emotions, they look for a glimmer of hope and look up to me with the trust that I as their provider will be able to “fix” all their concerns. I have felt the immense burden they carry and many times their emotions cross over and affect me too.
At that moment, the only thought racing through my mind was how could I decrease their physical and psychological distress to help them “feel better.” How could I decrease the burden they carry not just physically, but mentally and emotionally, and how could I empower them with the tools and strategies to build both physical and mental resilience?
The American Physical Therapy Association (APTA) supports interprofessional collaboration at the organizational and individual levels to promote research, education, policy, and practice in behavioral and mental health to enhance the overall health and well-being of society consistent with APTA’s vision. Physical, behavioral, and mental health are inseparably interconnected with overall health and well-being. It is within the professional scope of physical therapist practice to screen for and address behavioral and mental health conditions in patients, clients, and populations. This includes appropriate consultation, referral, or co-management with licensed health services providers in the prevention and management of behavioral and mental health conditions (1) through its position statement HOD P06-20-40-10.
The APTA also endorses evidence-based complementary and integrative interventions (HOD P06-18-17-47) underscores the importance of incorporating Integrative medicine practices within physical therapy. At the State level, as a member of the American Physical Therapy Association of New Jersey (2), I have been actively involved in Co-Charing the new Integrative Physical Therapy (IPT) Special Interest Group. IPT blends traditional physical therapy methods with holistic practices that address the whole person—physically, mentally, emotionally, and spiritually. This initiative aims to enrich therapists' capabilities in delivering Integrative Physical Therapy by leveraging a variety of methodologies, including Yoga, Mindfulness, Tai Chi, Hydrotherapy, Acupuncture, Acupressure, Zero Balancing, Reiki, Nutrition and Energy medicine to name a few.
According to the National Center of Complementary & Integrative Health (NCCIH), a branch of the National Institutes of Health (NIH), Mental health problems are common. In the United States, they affect about one-fourth of adults in any given year. According to the World Health Organization, mental illnesses account for more disability in developed countries than any other group of illnesses. Anxiety and mood disorders are the most common mental health problems. Researchers are investigating complementary and integrative health approaches for a variety of mental health problems, including anxiety & depression (3).
Anxiety disorders are one of the most common mental health concerns with a major contribution to the global burden of disease. Pharmacology and psychotherapy stand for the conventional treatment for anxiety disorders but these present limited efficacy, especially in the case of chronic anxiety, with high relapse rates often causing adverse side effects (4).
Anxiety is a natural part of the human experience, often serving as a protective mechanism in response to potential threats. However, when symptoms become chronic, disproportionate, or uncontrollable, anxiety can escalate into a clinical disorder. According to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, Text Revision (DSM-5-TR), anxiety disorders vary in presenting symptoms and may include excessive worry, panic attacks, restlessness, fatigue, difficulty concentrating, irritability, muscle tension, and sleep disturbances. Beyond these distressing psychological and emotional disturbances, the sequelae of anxiety extend into the realm of physical health. Research has pointed to a correlation between anxiety and increased pain responses, as well as the exacerbation of musculoskeletal disorders (5).
 Anxiety disorders have profound implications on both mental and physical health.
Anxiety disorders have profound implications on both mental and physical health.
There is growing interest in manual therapy modalities, with emerging research to alleviate related symptoms (5). A systematic review by West et al (2024) found that individuals receiving manual therapy interventions displayed a statistically significant reduction in anxiety intensity. The use of Acupressure for Anxiety is a common practice in Integrative Medicine, and the literature supports Acupressure as an effective and feasible alternative treatment for decreasing anxiety. A systematic review and meta-analysis published by He et al. (2019) found moderate evidence that Acupuncture and/or Acupressure was significantly associated with reduced cancer pain and decreased use of analgesics.
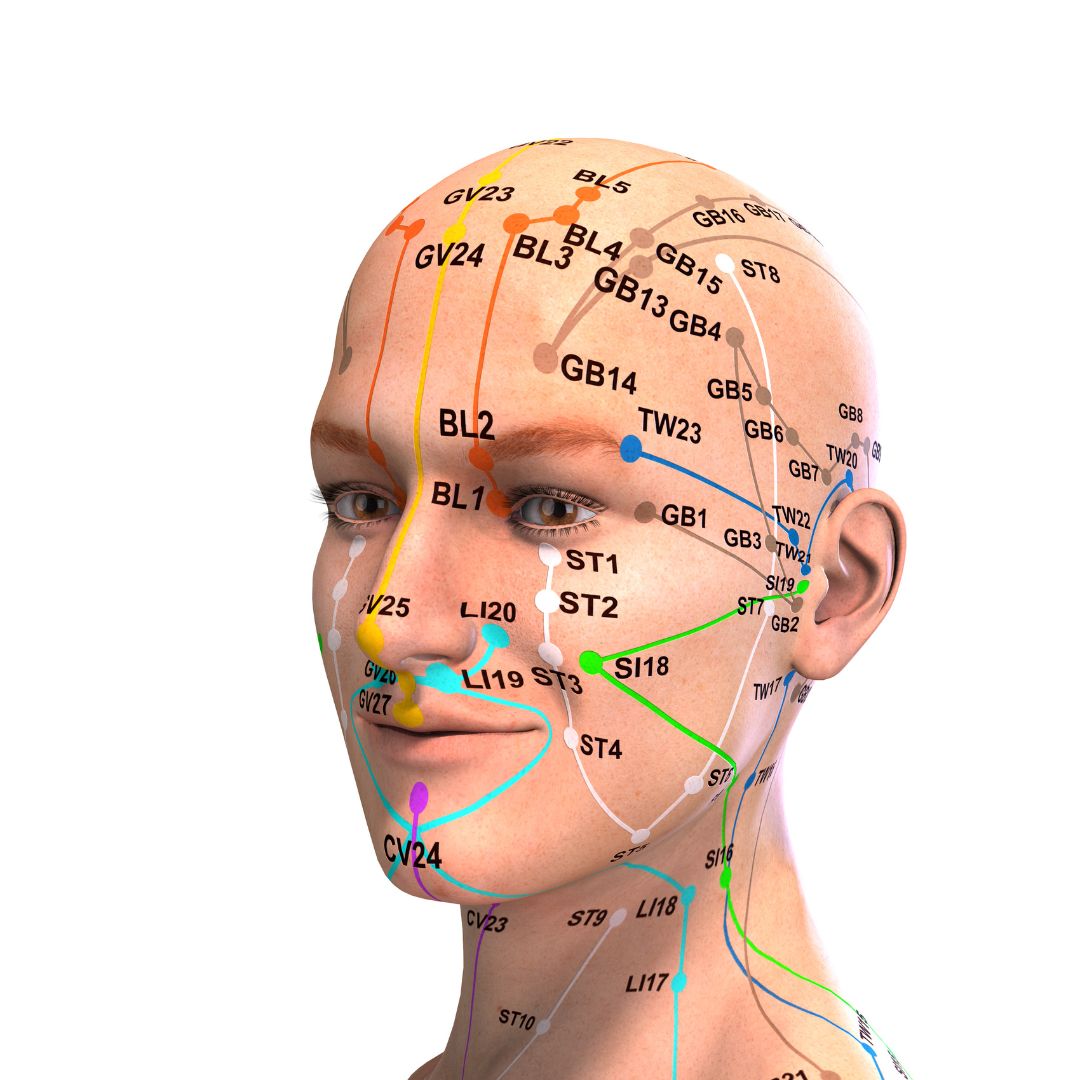
Acupressure is part of Traditional Chinese medicine (TCM), which is a comprehensive science that studies human health and disease. A central relational element of TCM is Yin and Yang, which refer to opposites, mutually exclusive yet complementary substances within the human body and the outside world. Based on the theory of the balance of Yin and Yang and the interaction of five elements (五行) (metal, wood, water, fire, and earth), TCM analyzes changes in the five Zang organs (五脏) (liver, heart, spleen, lung, and kidney), six Fu organs (六腑) (gallbladder, stomach, large intestine, small intestine, bladder, and triple energizer), extraordinary Fu-organs (奇恒之腑) (brain, marrow, bone, pulse, gallbladder, uterus, and ovary), meridians (the channels of basic substances transportation and connecting inside and outside of the human body), and qi-blood-body fluid (the basic substances constituting and maintaining the human life activities) (6).
A recent study by Yang et al (2021) cited several key Acupressure points that can help with Anxiety & fatigue (Heart 7, Spleen 6), chemotherapy-induced nausea, vomiting and anxiety in women with breast cancer ( Pericardium 6), primary dysmenorrhea ( Liver 3 ), anxiety & pain in cancer patients (Large Intestine 4, Heart 7), pre-operative cesarean section anxiety (Yintang EX-HN 3 and Heart 7), anxiety from the anticipation of surgery or treatment (Yintang EX-HN3, Heart 7 & Pericardium 6).
The literature also provides robust scientific evidence supporting Acupressure as an effective non-pharmacological therapy for the management of a host of conditions such as insomnia, chronic pelvic pain, dysmenorrhea, infertility, constipation, digestive disturbances, and urinary dysfunctions to name a few.
Case Study Clinically, our ability to target the nervous system to make a change in a patient’s pain and anxiety is extremely valuable from a pain neuroscience perspective. Recently a patient in her early 60s walked into the clinic with high anxiety and rectal pain rated 15/10. She reported that she spoke to her best friend and her friend who had found relief for anxiety with urinary retention with Acupressure had urged her to see me. She said, “I have tried everything, and you are my last hope!”
Clinically, our ability to target the nervous system to make a change in a patient’s pain and anxiety is extremely valuable from a pain neuroscience perspective. Recently a patient in her early 60s walked into the clinic with high anxiety and rectal pain rated 15/10. She reported that she spoke to her best friend and her friend who had found relief for anxiety with urinary retention with Acupressure had urged her to see me. She said, “I have tried everything, and you are my last hope!”
This patient had been to the ER twice over a span of 3 months and had undergone extensive testing with no diagnostic pathology. The only position she could find relief was in a side-lying but she was so distressed at the thought of “spending the rest of my life on the couch” that she refused to lie down in that position during the day, even if it meant relief from her excruciating pain.
The first thing I taught her was to notice how shallow her breath was, and how to use her breath deeply and actively to decrease her pain and anxiety by using two key Acupressure points Central Vessel 17 (CV 17), located at the center of her chest and Yintang EX-HN3, located between the eyebrows. CV17 is known to be a point for emotional healing while Yintang (EX-HN 3) is known to have a mentally stabilizing effect in Traditional Chinese Medicine (TCM). Within a few minutes, the patient felt very calm and relaxed and felt that she now had some control over her symptoms.
She was now able to focus and actively listen to me as I explained to her that we needed to work not just on the physical body, but also the mental, emotional, and energy body. I guided her to accept the “positional preference” of her body and “align” herself mentally and physically with what makes her “feel better.” So instead of feeling bad every time she thought about lying down to decrease her pain, I taught her to feel empowered that she had one position that she could use to break the cycle of her pain. This shift in how she approached her pain, as well as the potent Acupressure points to alleviate pain and anxiety, were a tremendous relief to her. I sent her home with a breathwork practice, a pelvic girdle stretching program, and an Acupressure program that targeted key Acupoints for self-regulation. The patient returned the next visit and reported that her pain was down from 15/10 to 5/10.
Over the course of the next few visits, using a combination of Acupressure as a self-regulation tool, targeted stretching of the lower back & pelvic girdle musculature, manual therapy, self-care, and breath work and awareness, this patient gained complete control over her anxiety and pain. She felt much calmer and empowered to have a physical self-regulation Acupressure practice which she could use to improve both her physical and mental health.
As a holistic pelvic health practitioner, who is psychologically informed with an Integrative physical therapy clinical practice, I recognize the deep-rooted mind-body connections and the need to address the “whole” person. In all the ancient and modern Energy healing practices, the breath is the key to working with the physical, mental, emotional, and the energy body. Acupressure is a powerful evidence-based energy healing practice that can be combined with breathing to heal the body in multiple dimensions. When a patient can “breathe better” and “feel better,” they can be steered easily towards building both physical and mental resilience.
As healthcare providers, we share this mental and emotional burden with our patients, and we must empower them with the tools and strategies to live healthy pain-free and anxiety-free lives.
To learn more about Acupressure, please join us for the upcoming remote course Acupressure for Optimal Pelvic Health scheduled for Feb 1st & 2nd. The course will introduce course participants to the basics of Traditional Chinese Medicine (TCM), Acupuncture & Acupressure. Of the 12 major Meridians or energy channels, this course will focus on the Bladder, Kidney, Stomach, and Spleen meridians. In addition, there are other important Meridian points that stimulate the nervous system and can be used for self-regulation to manage Anxiety, pain, and a host of other symptoms. The course also offers two potent Acupressure home exercise and wellness programs.
This course explores Yin yoga as a powerful holistic practice with Acupressure and will offer an evidence-based perspective on how Yin poses within each meridian can channelize energy through neurodynamic pathways with powerful integrative applications to facilitate healing in multiple dimensions.
References
- The Role of the Physical Therapist and the American Physical Therapy Association in Behavioral and Mental Health: Position statement https://www.apta.org/apta-and-you/leadership-and-governance/policies/role-pt-apta-behavioral-mental-health
- American Physical Therapy Association of New Jersey: Integrative Physical Therapy Special Interest Group https://aptanj.org/page/IPTSIG
- National Center of Complementary & Integrative Health (NCCIH) https://www.nccih.nih.gov/health/anxiety-and-complementary-health-approaches
- Amorim D, Amado J, Brito I, et al. Acupuncture and electroacupuncture for anxiety disorders: A systematic review of the clinical research. Complement Ther Clin Pract. 2018;31:31-37. doi:10.1016/j.ctcp.2018.01.008
- West KL, Huzij T. A systematic review of manual therapy modalities and anxiety. J Osteopath Med. 2024;124(11):487-497. Published 2024 Jun 24. doi:10.1515/jom-2024-0001
- Yang J, Do A, Mallory MJ, Wahner-Roedler DL, Chon TY, Bauer BA. Acupressure: An Effective and Feasible Alternative Treatment for Anxiety During the COVID-19 Pandemic. Glob Adv Health Med. 2021;10:21649561211058076. Published 2021 Dec 12. doi:10.1177/21649561211058076
- He Y, Guo X, May BH, et al. Clinical Evidence for Association of Acupuncture and Acupressure With Improved Cancer Pain: A Systematic Review and Meta-Analysis. JAMA Oncol. 2020;6(2):271-278. doi:10.1001/jamaoncol.2019.5233
- Zhang SQ, Li JC. An introduction to traditional Chinese medicine, including acupuncture Anat Rec (Hoboken). 2021;304(11):2359-2364. doi:10.1002/ar.24782
- Abbott R, Hui EK, Kao L, et al. Randomized Controlled Trial of Acupressure for Perception of Stress and Health-Related Quality of Life Among Health Care Providers During the COVID-19 Pandemic: The Self-Acupressure for Stress (SAS) Trial. Am J Med Open. 2023;10:100056. doi:10.1016/j.ajmo.2023.100056
- Chen SR, Hou WH, Lai JN, Kwong JSW, Lin PC. Effects of Acupressure on Anxiety: A Systematic Review and Meta-Analysis. J Integr Complement Med. 2022;28(1):25-35. doi:10.1089/jicm.2020.0256
- Monson E, Arney D, Benham B, et al. Beyond Pills: Acupressure Impact on Self-Rated Pain and Anxiety Scores. J Altern Complement Med. 2019;25(5):517-521.
Author Bio
Rachna Mehta PT, DPT, CIMT, OCS, PRPC, RYT 200
 Rachna Mehta PT, DPT, CIMT, OCS, PRPC, RYT 200 (she/her) graduated from Columbia University, New York with a Doctor of Physical Therapy degree. Rachna has since been working in outpatient hospital and private practice settings with a dual focus on Orthopedics and Pelvic Health. She was instrumental in starting one of the first Women’s Health Programs in an outpatient orthopedic clinic setting in Mercer County, New Jersey in 2009. She has authored articles on pelvic health for many publications. She is a Certified Integrated Manual Therapist through Great Lakes Seminars, is Board-certified in Orthopedics, is a certified Pelvic Rehab Practitioner, and is also a registered yoga teacher through Yoga Alliance. Rachna has trained in both Hatha Yoga and Yin Yoga traditions and brings the essence of Yoga to her clinical practice.
Rachna Mehta PT, DPT, CIMT, OCS, PRPC, RYT 200 (she/her) graduated from Columbia University, New York with a Doctor of Physical Therapy degree. Rachna has since been working in outpatient hospital and private practice settings with a dual focus on Orthopedics and Pelvic Health. She was instrumental in starting one of the first Women’s Health Programs in an outpatient orthopedic clinic setting in Mercer County, New Jersey in 2009. She has authored articles on pelvic health for many publications. She is a Certified Integrated Manual Therapist through Great Lakes Seminars, is Board-certified in Orthopedics, is a certified Pelvic Rehab Practitioner, and is also a registered yoga teacher through Yoga Alliance. Rachna has trained in both Hatha Yoga and Yin Yoga traditions and brings the essence of Yoga to her clinical practice.
Rachna currently practices in an outpatient setting. Her clinical practice has focused on an Integrative physical therapy approach blending traditional physical therapy methods with holistic practices that address the whole person - physically, mentally, emotionally, and spiritually. She specializes in working with pelvic health patients who have bowel and bladder issues with high pelvic pain which sparked her interest in Eastern holistic healing traditions and complementary medicine. She has spent many hours training in holistic healing workshops with teachers based worldwide. She is a member of the American Physical Therapy Association and a member of APTA’s Academy of Orthopaedic Physical Therapy and the Academy of Pelvic Health Physical Therapy.
Rachna also owns TeachPhysio, a PT education and management consulting company. Her course Acupressure for Optimal Pelvic Health brings a unique evidence-based approach and explores complementary medicine as a powerful tool for holistic management of the individual as a whole focusing on the physical, emotional, and energy body.

One of the things you'll learn when working with the pediatric population is that they're a different species than adults. Some of the common diagnoses may overlap, such as chronic constipation, fecal smearing, or nocturia. The way they present may be similar and the treatment ideas and philosophy may also overlap. However, how these treatments are implemented may vary when treating a child versus an adult. When I think of treating the pediatric population, I like to divide my thought process and approach into five different groups.
To me, pediatrics can be babies/infants, toddlers, preschool-aged children, elementary school-aged children, and then tweens/teens. It may feel excessive to divide this population into so many subgroups, but each of those groups has its own treatment considerations based on the child's development at that time in their lives. An infant may be working on regulating their GI system as they transition from being in-utero to being born. Toddlers and school-aged children are working through various internal and social benchmarks to help them continue to grow and develop. Likewise, a tween or teen may be managing the changes to their genitourinary system as they work through puberty towards adulthood or grappling with their sexuality and sexual identity as they mature.
Regardless of what stage they are in life, patients will potentially present differently and also need a customized treatment plan to meet them where they are at. When I have colleagues jumping into pediatric pelvic health, I rarely worry they have the clinical skills to help with the physical and physiological challenges a child may face. The place I see many clinicians struggle is how to apply the skills they know to these tiny humans in front of them. Treating reflux in an eighty-year-old versus an 8-week old person is very different.
Children can't always tell us what they're feeling and how they're feeling it. An adult that has the ability for interoception can tell us what they're feeling, how they're feeling it, and how our treatment interventions are affecting their condition and goals. A pediatric patient may not be able to provide this feedback so subtle signs such as skin color, facial expressions, and body language may be your only clues. Likewise, being able to interpret a baby or child's negative reactions, such as crying, trying to get away, trying to hide, or otherwise avoiding therapy is a very necessary skill when working with pediatrics. Long story short, we can ask an adult if what we're doing is working, with a pediatric patient, we have to be more creative in determining our efficacy.
Children can't give consent. Children can give assent, meaning that they will comply with what you are asking them to do. This can feel tricky and if you struggle with this I recommend taking Ethics Considerations for Pediatric Pelvic Health on July 27, 2025. When working with pediatric patients, the take-home point is that the therapist will always have more power than the child that they are working with. This means that the therapist has to be very careful in how they wield their power dynamic, to be most beneficial and fair to the child.
Children usually can't be autonomous with their home programs. When I think about home programs, some of my adult clients even struggle to complete these correctly or regularly. Many times, especially when they're under the age of 10 years old, children cannot be autonomous with their home programs. This means that a practitioner has to consider what is going on in the home, the living situation that the child is in, their support system, the financial resources available to the child, and other factors that may act as help or hindrance to their home program activation.
Children need caregiver support and guidance. As we said above, most of the care that we're giving to a child in the home is going to be provided with the support or completely by a parent or caregiver. We have to make sure that the child's guardian is on board with the treatment plan, has the resources and ability to enact the treatment plan, and is being respectful to the child as they work on the program at home. As therapists, we have to help navigate bumps and challenges on this road to recovery for the child and their support system, or else we're not doing all aspects of our job. I am always asking myself questions like:
“Can this family afford this?”
“Do they have time for this?”
“Do they understand why I am asking them to do this?”
“Is me asking this of this family adding unnecessary stress to this child or the caregivers?”
Children deserve to be offered to “buy in” to their plan of care. One of the mistakes I see colleagues make is understanding the wisdom of children. Yeah, they're tiny. Yes, they sometimes eat their boogers and think poop jokes are funny. Still, children are much more intuitive than we frequently give them credit for. I've had kiddos as little as 3 years old be excited to drink their “poop juice” to help move their “poop train.” If we “make it make sense” for them, they become the biggest, most powerful part of their care team! Teaching lifelong good habits is something I know most therapists love about their jobs and working with kids provides this so easily.
If you're a provider who is jumping from adults to pediatrics or if you are new to pelvic floor with pediatrics in general, Pediatric Pelvic Floor Playskills is a class that walks you through challenges and solutions by age, sample treatment plans, and problem-solving case studies to build your confidence and efficacy in treating this population. Hope you decide to come play with us on January 25th!
AUTHOR BIO:
Mora Pluchino, PT, DPT, PRPC

Mora Pluchino, PT, DPT, PRPC (she/her) is a graduate of Stockton University with a BS in Biology (2007) and a Doctorate of Physical Therapy (2009). She has experience in a variety of areas and settings, working with children and adults, including orthopedics, bracing, neuromuscular issues, vestibular issues, and robotics training. She began treating Pelvic Health patients in 2016 and now has experience treating women, men, and children with a variety of Pelvic Health dysfunction. There is not much she has not treated since beginning this journey and she is always happy to further her education to better help her patients meet their goals.
She strives to help all of her patients return to a quality of life and activity that they are happy with for the best bladder, bowel, and sexual functioning they are capable of at the present time. In 2020, She opened her own practice called Practically Perfect Physical Therapy Consulting to help meet the needs of more clients. She has been a guest lecturer for Rutgers University Blackwood Campus and Stockton University for their Pediatric and Pelvic Floor modules since 2016. She has also been a TA with Herman & Wallace since 2020 and has over 150 hours of lab instruction experience. Mora has also authored and instructs several courses for the Institute.