Running is a sport that spans a whole gamut of activity levels. Some runners run a mile or two casually every evening, some sprint competitively, and some runners train towards running marathons.
Female runners of all ages report urinary incontinence while running. The incontinence that is reported while running is not restricted to occurring while running at high speeds or long distances. There are runners who complain of leakage as soon as they start running, while there are others who leak mid-way into the run. Part of treating pelvic floor dysfunction in runners involves the ability to be able to tease out where the breakdown that is causing the dysfunction is occurring. Clinicians who treat pelvic dysfunction in runners have to be able to perform a running analysis to find the biomechanical faults that can contribute to the dysfunction that the runner is experiencing.
One common fault found during a running analysis is a pelvic hip drop on the stance leg, possibly indicating a weakness in the hip abductor muscles, specifically the gluteus medius.Hip abductor muscle weakness has been found to be a contributing factor in many running-related injuries, including iliotibial band syndrome, patellofemoral pain syndrome, stress fractures, and Achilles tendinopathy.(1) But, one can also link weakness in the hip abductors to increased pelvic floor dysfunction in runners
There are myofascial connections found through magnetic resonance imaging between the gluteal muscles and the levator ani and fossa ischioanalis. Multiple studies have found a significant correlation between weakness in the hip abductors, external rotators, stress urinary incontinence, and increased urinary urgency and frequency.(2,3)
Understanding the relationship between hip strength, biomechanics, and pelvic floor function is essential in supporting runners who experience urinary incontinence. By identifying and addressing underlying factors such as hip abductor weakness and faulty running mechanics, clinicians can help reduce leakage, improve overall running efficiency, and decrease risk of injury. With targeted assessment and treatment strategies, runners can return to the sport they love with greater confidence, strength, and pelvic health.
In the Running and Pelvic Health course taught by Aparna Rajagopal and Leeann Taptich, there will be instruction on how to perform a running analysis, how to assess for hip abductor weakness, and how to treat those findings to decrease pelvic floor dysfunction in runners. Please join us on November 1, 2025, for our next course.
Resources
- Mucha MD, Caldwell W, Schlueter EL, Walters C, Hassen A. Hip abductor strength and lower extremity running-related injury in distance runners: A systematic review. Journal of Science and Medicine in Sport. 2017; 20: 349-355.
- Marques SAA, da Silverira SRB, Passaro AC, Haddad JM, Baracat EC, Ferreira EAG. Effect of pelvic floor and hip muscle strengthening in the treatment of stress urinary incontinence: A randomized clinical trial. J Manipulative Physiol Ther. 2020; 43(3): 247-256.
- Foster SN, Spitznagle TM, Tuttle LJ, Sutcliffe S, Steger-May K, Lowder JL, Meister MR, Ghetti C, Wang J, Mueller MJ, Harris-Hayes M. Hip, and pelvic floor muscle strength in women with and without urgency and frequency predominant lower urinary tract symptoms. J Womens Health Phys Therap. 2021; 45(3): 126-134.
AUTHOR BIO
Leeann Taptich DPT, SCS, MTC, CSCS
 Leeann Taptich, DPT, SCS, MTC, CSCS (she/her) has been a physical therapist since 2006. She graduated with a BS in Kinesiology from Michigan State University and a Doctorate of Physical Therapy from the University of St Augustine. In 2009, she earned her Manual Therapy from the University is St Augustine and her board certification as a Sports Certified Specialist in 2018.
Leeann Taptich, DPT, SCS, MTC, CSCS (she/her) has been a physical therapist since 2006. She graduated with a BS in Kinesiology from Michigan State University and a Doctorate of Physical Therapy from the University of St Augustine. In 2009, she earned her Manual Therapy from the University is St Augustine and her board certification as a Sports Certified Specialist in 2018.
Leeann leads the Sports Physical Therapy team at Henry Ford Macomb Hospital in Michigan, where she mentors a team of therapists. She also works very closely with the pelvic team at the hospital, which gives her a very unique perspective of the athlete. With her combination of credentials and her exposure to pelvic health, she is able to use a very eclectic but complete approach in her treatment of orthopedic and sports patients. With the hospital system, she is involved with the community promoting health and wellness at local running competitions and events.
Leeann is passionate about educating and teaching and has assisted in teaching multiple courses at the local State university PT department. She is co-chair of the continuing education committee at her hospital, where she writes and develops courses. She is co-author of the Breathing and Diaphragm class at Herman & Wallace.
Leeann lives in the metro Detroit area with her husband and two children. She enjoys hiking, traveling, and watching football.

Pelvic organ prolapse (POP) and stress urinary incontinence (SUI) are common conditions affecting people with a vagina across the lifespan, often leading to significant impairments in quality of life. Conservative management is increasingly prioritized, particularly among those who are not surgical candidates, wish to delay surgery, or are managing symptoms during pregnancy or postpartum recovery. Pessaries, intravaginal devices used to support the pelvic organs or compress the urethra, represent a cornerstone of conservative pelvic health management. Their safe, relatively non-invasive nature and growing evidence base make them essential tools in a clinician’s pelvic health toolkit. Pessaries are considered relatively safe for many patient populations and are well tolerated when a proper fit is achieved (Rogers 2017). Well fit pessaries are expected to allow people with POP to participate in a full range of physical activities and exercise comfortably (O’Dell 2012).
This essay explores the current evidence on pessary use, including types, indications, contraindications, patient selection, fitting considerations, and integration with pelvic therapy. Emphasis is placed on empowering pelvic health providers to incorporate pessaries confidently and collaboratively in clinical practice.
Clinical Indications
 Pessaries can be used for a range of pelvic health conditions, including:
Pessaries can be used for a range of pelvic health conditions, including:
- Pelvic organ prolapse (POP), this includes management of symptomatic cystocele, rectocele, uterine prolapse, or vault prolapse.
- Stress urinary incontinence (SUI): Especially in cases where the patient is participating in pelvic therapy and making gains in pelvic floor function but continues to have symptoms during lifting or during high-impact activities.
- Pregnancy-related prolapse or urinary symptoms: Providing support during gestation. This is prescribed by a birth provider including an MD, NP, or CNM.
- Postpartum recovery: For temporary support during tissue remodeling.
Types of Pessaries and Their Functions
 Pessaries are broadly categorized into supportive and space-filling devices. The selection of type depends on the specific condition being treated, the degree of prolapse, and the patient’s anatomy and functional goals.
Pessaries are broadly categorized into supportive and space-filling devices. The selection of type depends on the specific condition being treated, the degree of prolapse, and the patient’s anatomy and functional goals.
- Support Pessaries
These include the ring pessary (with or without support) and are generally used for mild to moderate prolapse or stress urinary incontinence. Ring with support is frequently used in patients with a widened introitus some apical loss of support (either the cervix or the cuff in the event of hysterectomy). Ring without support can be a good choice for patients with mild to moderate cystocele and/or rectocele with generally good pelvic floor muscle tone.
- Space-Filling Pessaries
Designed for more advanced prolapse or when support pessaries fail, these include Gellhorn, Donut, and Cube pessaries. The Gellhorn is often used in cases of more severe prolapse (stage III or IV prolapse). For stress urinary incontinence, the incontinence dish or ring with knob can elevate the urethra and reduce leakage under load of the knob.
Contraindications and Cautions
Although pessaries are safe, contraindications and red flags must be observed. They include:
- Active vaginal infections (e.g., candidiasis or bacterial vaginosis) should be treated prior to fitting.
- Severe vaginal atrophy may increase the risk of erosion and should be managed with topical estrogen before long-term use.
- Noncompliance with prior use and cleaning, or cognitive impairment may limit self-care ability unless caregivers are trained.
- Allergy to pessary material (e.g., latex, silicone), though rare, must be considered.
Fitting Process: A Collaborative and Individualized Approach
 Proper fitting is essential for comfort, function, and adherence. The process typically includes a pelvic exam to assess prolapse stage, vaginal length, introital laxity, and tissue integrity. Additionally, trial and error during fitting with a range of pessary sizes and types is common. The right pessary should sit comfortably behind the pubic symphysis and under the cervix or vaginal vault without protruding or causing discomfort.
Proper fitting is essential for comfort, function, and adherence. The process typically includes a pelvic exam to assess prolapse stage, vaginal length, introital laxity, and tissue integrity. Additionally, trial and error during fitting with a range of pessary sizes and types is common. The right pessary should sit comfortably behind the pubic symphysis and under the cervix or vaginal vault without protruding or causing discomfort.
Functional testing during the fitting process is helpful to ensure comfort and optimal use for the patient. To do this, ask the patient to stand, bear down, cough, and walk around the office to ensure stability and effectiveness.
Patient education: On insertion, removal, cleaning, and signs of complications.
Follow-up after a pessary is fit is usually done after 1–2 weeks, then at 3-month intervals depending on patient autonomy and tissue health. Patients should also be instructed in managing complications and problem solving. Most pessary complications are minor and manageable, including:
- Increased vaginal discharge
- Odor or irritation
- Light bleeding or spotting, especially in estrogen-deficient tissues
- Expulsion or shifting of the pessary
- Urinary retention (rare, more common with space-filling devices)
Management strategies:
- Encourage regular removal and cleaning (daily to weekly for self-managing patients; every 1–3 months for clinician-managed).
- Use vaginal estrogen for tissue health in postmenopausal patients.
- Resize or switch device types if discomfort or expulsion occurs.
- Refer for evaluation to a physician or nurse practitioner if persistent bleeding, pain, or infection is noted.
Patient Empowerment and Adherence
Patient-centered education improves long-term success with a pessary. Education that pessaries are not permanent and can be removed at any time often alleviates anxiety that they may be feeling. Additionally, pessaries do not stretch or weaken the vagina; in fact, they often support pelvic rehabilitation efforts in conjunction with pelvic health therapy. Additionally, self-care is safe and achievable with practice and symptom relief can be immediate, as the patient works on restoring mobility, confidence, and function. Offering video tutorials, printed guides, and in-person instruction increases adherence and reduces fear or confusion.
Conclusion
Pessaries are a powerful, evidence-based tool for managing pelvic organ prolapse and stress urinary incontinence. When thoughtfully integrated into a multidisciplinary pelvic health plan, they provide a non-invasive, low-risk option that empowers patients while supporting tissue healing and function. Pelvic health providers play a crucial role in identifying appropriate candidates, facilitating fittings, and supporting ongoing care, especially when combined with targeted physical therapy. Understanding the nuances of pessary selection, education, and monitoring will continue to elevate care standards and improve outcomes in this essential area of women’s health.
References:
- O’Dell K, Atnip S. (2012). Pessary Care: Follow Up and Management of Complications. Urologic Nursing.32 (3), 126-145.
- Rogers, R. G., & Fashokun, T. B. (2017). Pelvic organ prolapse in women: Epidemiology, risk factors, clinical manifestations, and management. Waltham, MA: Wolters Kluwer.
AUTHOR BIO
Dr. Amanda Olson
 Dr. Amanda Olson is a pelvic health physical therapist, entrepreneur, author, and global educator with a passion for empowering men and women and advancing conversations around pelvic health. She has dedicated her career to breaking stigmas around pelvic health, supporting patients with conditions including cancer, incontinence, pelvic pain, cancer, pelvic organ prolapse, endometriosis, and MRKH, and developing innovative solutions to improve quality of life.
Dr. Amanda Olson is a pelvic health physical therapist, entrepreneur, author, and global educator with a passion for empowering men and women and advancing conversations around pelvic health. She has dedicated her career to breaking stigmas around pelvic health, supporting patients with conditions including cancer, incontinence, pelvic pain, cancer, pelvic organ prolapse, endometriosis, and MRKH, and developing innovative solutions to improve quality of life.
Dr. Olson is the president of Intimate Rose, a successful pelvic health device company that includes tools for managing pelvic pain, holds multiple patents, and has authored six peer-reviewed journal articles as well as the book Restoring the Pelvic Floor for Women. She is also the recipient of the Elizabeth Nobel Award, the highest honor bestowed in the field of pelvic health. She has collaborated with clinics worldwide, transforming lives through her tools, teaching, and consulting. As an advocate for patients with sensitive issues, and medical innovation, Dr. Olson is committed to bridging the gap in pelvic and women’s healthcare and bringing education to underserved patient populations.

Kelly Sammis, PT, OCS, CLT, AFDN-S is a physical therapist, educator of dry needling and all things pelvic, Pilates instructor, wife, and mama living and working in Parker, Colorado. She specializes in the treatment of male and female pelvic floor dysfunction, athletic injury/return to sport, sports performance, and persistent pain. Her formal education took place at Ohio University (2007) and The University of St Augustine for Health Sciences (2010). Kelly serves as the lead faculty developing and teaching dry needling and pelvic health courses nationwide. Kelly co-instructs the Herman & Wallace Dry Needling courses along with site fellow faculty member Tina Anderson, MS PT.
Urinary incontinence (UI) is defined as ‘any complaint of involuntary leakage or urine’ that has several different subtypes based on when this leakage occurs1. UI is a common and relevant condition that has a profound influence on well-being and quality of life of many patients worldwide. Millions of men and women throughout the world are affected. According to our body of evidence, UI can affect anywhere between 5-70% of the female population2-4 and 11-32% of the male population5,6, contributing to decreased participation in preferred daily, work and recreational activities alongside an immense economic burden for some of those affected.1-7These symptoms have not only been shown to have a significant impact on a person’s quality of life, but also on their mental health status.7
While UI is both common and very bothersome, it is also very treatable. I would love nothing more than to see our society and healthcare continuum recognize that UI is something that is ABNORMAL versus the typical categorization that it is a normal part of the aging, postpartum or postoperative experience.
Common, not normal. Common, but treatable.
UI can be treated with lifestyle and behavioral interventions, bladder training, electrical stimulation, pelvic floor muscle training (PFMT) with or without biofeedback, physical therapy, neuromodulation, periurethral injections and, in some cases, surgical intervention. 5,8
Understanding the continence mechanism
In a well-functioning pelvic floor, the connective tissue of the ligaments and fascia act together with the pelvic floor musculature to counteract the impact of any increase in intra-abdominal pressure and ground reaction forces, helping to maintain our continence.8-10 This is an automatic function, requiring no need to think about voluntary contraction of the pelvic floor musculature. 8 When this mechanism is not working adequately, which can undoubtedly be multifactorial, urinary incontinence may occur.
The external urethral sphincter mechanism is a complex system of striated muscle which includes fiber blends from the urogenital triangle musculature and the anterior muscle bundle of the levator ani. 11 The good news here is that we, as rehabilitative clinicians, have many different tissue targets for treatment of UI.
Dry Needling and UI
Dry needling (DN) encompasses the insertion of solid filament, non-injectate needles into, alongside or around muscles, nerves or connective tissues with or without mechanical and/or electrical stimulation for the management of pain and dysfunction in neuromusculoskeletal conditions. DN is both effective and efficient in modulating the central and peripheral nervous systems as well as the somatic tissues, including the pelvic floor.
There is a growing body of evidence that has provided us with an understanding on how to best utilize this technique in our clinical practice as it relates to UI.12-17 With the external urethral sphincter and associated tissues being a main player in our urinary continence mechanism, it provides a road map on how we can utilize DN to treat UI. This boils down to two things: (1) tissue specificity and (2) utilization of electrical stimulation. DN provides us with an avenue to directly influence a specific tissue as we are able to use an indwelling needle electrode placed strategically into a muscular or perineural tissue target. Using that tactically placed indwelling electrode we can then precisely deliver electrical stimulation, essentially speaking the language of the neuromotor system, making this technique one of the most effective tools we have as rehabilitative clinicians to treat UI.
Ultimately, we are able to stimulate the pudendal nerve alongside the targeted tissues. This can help to improve electric activation, proprioception and coordination in pelvic floor contraction during situations that contribute to UI.18,19,22 Additionally, the pudendal nerve is an efferent nerve for the external urethral sphincter, so this treatment is capable of increasing the pressure of urethral closure, improving UI. Another important factor is that electrical stimulation has been shown to increase blood flow to the urethra and pelvic floor musculature, lending towards improvements in neuromuscular connections, muscle fiber function and genital atrophy, all leading to improvements in the mechanism of urethral closure. 19-22
The power of the tissue reset that DN provides has changed clinical outcomes for the better. It has, and will continue to, positively impact and change the lives of many patients through facilitating a more balanced homeostatic baseline within the tissues, healthier motor recruitment patterns and optimal neuromuscular utility to re-establish function. Want to add this tool to your clinical practice? Check out our Dry Needling course offerings with Herman & Wallace!
References:
- Haylen BT, de Ridder D, Freeman RM, et al. An International Urogynecological Association (IUGA)/ International Continence Society (ICS) joint report on the terminology for female pelvic floor dysfunction. Int Urogynecol J. 2010;21:5–26
- Milson I and Gyhagen M. The prevalence of urinary incontinence. Climacteric. 2019;22(3):217-222
- Carryer, J, Weststrate, J, Yeung, P et al. Prevalence of key care indicators of pressure injuries, incontinence, malnutrition, and falls among older adults living in nursing homes in New Zealand. Research In nursing & Health. 2017;40(6):555–563
- Damian, J, Pastor-Barriuso, R, Garcia Lopez, FJ et al. Urinary incontinence and mortality among older adults residing in care homes. Journal of Advanced Nursing. 2017;73(3):688–699
- Gacci M, Sakalis VI, Karavitakis M et al. European Association of Urology guidelines on male urinary incontinence. European Urology. 2022;82:387-398
- Cao C, Zhang C, Sriskandarajah C et al. Trends and racial disparities in the prevalence of urinary incontinence among men in the USA, 2001-2020. European Urology Focus. 2022; https://doi.org/10.1016/j.euf.2022.04.015
- Krhut J, Gartner M, Mokris J et al. Effect of severity of urinary incontinence on quality of life in women. Neurourol Urodyn. 2018;37:1925–1930
- Bo K. Physiotherapy management of urinary incontinence in females. Journal of Physiotherapy. 2020;
- Ashton-Miller J, DeLancey JOL. Functional anatomy of the female pelvic floor. In: Bø K, Berghmans B, Van Kampen M, Mørkved S, eds. Evidence based physical therapy for the pelvic floor. Bridging science and clinical practice. Chapter 3. Edinburgh: Elsevier; 2015:19–34
- DeLancey JOL, Low LK, Miller JM et al. Graphic integration of causal factors of pelvic floor disorders: an integrated life span model. Am J Obstet Gynecol. 2008;199:610.e1–610.e5
- Suriyut J, Muro S, Baramee P et al. Various significant connections of the male pelvic floor muscles with special reference to the anal and urethral sphincter muscles. Anatomincal Science Internatiional. 2020;95:305-312
- Feng X, Lv J, Li M et al. Short-term efficacy and mechanism of electric pudendal nerve stimulation versus pelvic floor muscle training plus transanal electrical stimulation in treating post-radical prostatectomy urinary incontinence. Oncology. 2022;160:168-175
- Wang S, Zhang S. Simultaneous perineal ultrasound and vaginal pressure measurement prove the action of electrical pudendal nerve stimulation in treating female stress incontinence. BJU Int. 2012;110:1338–1343
- Wang S, Lv J, Feng X, Wang G, Lv T. Efficacy of electrical pudendal nerve stimulation in treating female stress incontinence. Urology. 2016;91:64–69
- Wang S, Lv J, Feng X, Lv T. Efficacy of electrical pudendal nerve stimulation versus transvaginal electrical stimulation in treating female idiopathic urgency urinary incontinence. J Urology. 2017;197:1496–1501
- Wang S, Zhang S, Zhao L. Long-term efficacy of electric pudendal nerve stimulation for urgency-frequency syndrome in women. International Urogynecology Journal. 2014;25:397-402.
- Li T, Feng X, Lv J et al. Short-term clinical efficacy of electric pudendal nerve stimulation of neurogenic lower urinary tract disease: a pilot research. Urology. 2018;112:69-73
- Monga AK, Tracey MR, Subbaroyan J. A systematic review of clinical studies of electrical stimulation for treatment of lower urinary tract dysfunction. Int Urogynecol J. 2002;23:993–1005
- Chai TC, Steers WD. Neurophysiology of micturition and continence in women. Int Urogynecol Urol. 1997;8:85–97
- Spruijt J, Vierhout M, Verstraeten R, et al. Vaginal electrical stimulation of the pelvic floor: a randomized feasibility study in urinary incontinent elderly women. Acta Obstet Gynecol Scand. 2003;82:1043–8
- Balcom AH, Wiatrak M, Biefeld T et al. Initial experience with home therapeutic electrical stimulation for continence in myelomenin-gocele population. J Urol. 1997;158:1272–6
- Correia GN, Pereira VS, Hirakawa HS et al. Effects of surface and intravaginal electrical stimulation in the treatment of women with stress urinary incontinence: randomized controlled trial. Euro J of Ob & Gyn and Reproductive Bio. 2014;173:113-118
Dry Needling and Pelvic Health - Live Course

Course Dates:
Worcester, MA - November 12-13, 2022
Katy, TX - January 21-22, 2023
Birmingham, AL - September 16-17, 2023
Price: $995
Experience Level: Beginner
Contact Hours: 26
Description: This is the foundational dry needling course in our three course pelvic health series. Practitioners will learn an innovative approach to treating clients with pelvic floor and neuromusculoskeletal dysfunction commonly associated with lumbopelvic pain, incontinence, voiding dysfunction and/or sexual pain or dysfunction. This foundational dry needling course will instruct participants in the application of dry needling to female pelvic floor musculature and associated neuroanatomical structures including the lumbosacral spine, abdomen, pelvis, and hip joint complex. This course will provide a comprehensive review of anatomy, a strong emphasis on safety and precautions, ample lab time to optimize dry needling techniques, as well as dialogue surrounding clinical integration and relevant evidence.
Dry Needling and Pelvic Health: Advanced Concepts and Neuromodulation - Live Course

Course Dates:
Salt Lake City, UT - November 5-6, 2022
Price: $995
Experience Level: Advanced
Contact Hours: 26
Description: Dry Needling and Pelvic Health: Advanced Concepts and Neuromodulation is a lab intensive, hybrid course designed with the pelvic health practitioner in mind. This course is an innovative approach to treating clients with pelvic floor dysfunction commonly associated with pelvic pain, incontinence, voiding dysfunction and/or sexual pain or dysfunction. This advanced dry needling course will instruct participants in the application of dry needling to female and male pelvic floor musculature and associated neuroanatomical structures including the thoracolumbar spine, trunk, abdomen, pelvis and hip joint complex. This course will also discuss and integrate the concept of and techniques associated with neuromodulation. Additionally, there will be a comprehensive review of anatomy, a strong emphasis on safety and precautions, ample lab time to optimize dry needling techniques, as well as dialogue surrounding clinical integration and relevant evidence.
Tibial nerve stimulation has been shown in the literature to be effective for individuals experiencing idiopathic overactive bladder in randomized controlled trials. A systematic review was performed by Schneider, M.P. et al. in 2015 looking at safety and efficacy of its use in neurogenic lower urinary tract dysfunction. Many variables were examined in this review, which included 16 studies after exclusion. The review looked at:

- Acute stimulation (used during urodynamic assessment only)
- Chronic stimulation (6-12 weeks of daily-weekly use)
- Percutaneous or transcutaneous (frequencies, pulse widths, perception thresholds, durations)
- Urodynamic parameter changes baseline to post treatment
- Post void residual changes
- Bladder diary variables
- Patient adherence to tibial nerve stimulation
- Any adverse events
The exact mechanism of these types of neuromodulation stimulation procedures remains unclear, however it does appear to play a role in neuroplastic reorganization of cortical networks via peripheral afferents. No specific literature is currently available for the mechanism on action related to neurogenic lower urinary tract dysfunction. Different applications of neuromodulation however have been studied in the neurogenic populations.
One of the randomized controlled trials they report on included 13 people with Parkinson disease. The researchers looked at a comparison between the use of transcutaneous tibial nerve stimulation (n = 8) and sham transcutaneous tibial nerve stimulation (n=5). Transcutaneous tibial nerve stimulation (TTNS) or sham stimulation was delivered to the people with Parkinson disease 2x/week for 5 weeks, 30-minute sessions (10 total sessions). Unilateral electrode placement was utilized, first electrode applied below the left medial malleolus and second electrode 5 cm cephalad. Confirmation of placement was obtained with left great toe plantar flexion. It is important to note the use of the stimulation intensity is reduced to below the motor threshold during the active treatment to direct the stimulation via peripheral afferents.
Urodynamic testing was performed at baseline and post treatment and revealed statistically significant differences with greater volumes at strong desire and urgency in the TTNS group. Additionally, the TTNS group experienced a 50% reduction in nocturia whereas in the sham group nocturia frequency remained the same. A three-day bladder diary completed by each of the groups also revealed significant positive changes in frequency, urgency, urge urinary incontinence and hesitancy only in the TTNS group.
Conservative management of neurogenic bladder in populations such as Parkinson disease is very important. These individuals experience lower quality of life ratings related to lower urinary tract dysfunction, higher risk of falling with needs to rush to the bathroom, their caregivers experience a higher level stress and burden of care, and tolerance to anticholinergic medications is very poor with multiple unwanted side effects that compound and worsen other symptoms that might be present from the disease process.
Please join us for Neurologic Conditions and Pelvic Floor Rehab to learn how you can help your patients using this modality as one option. Participate in a lab session to learn electrode placement and other parameters to achieve best clinical results for your patients.
1. Perissinotto, M. C., D'Ancona, C. A. L., Lucio, A., Campos, R. M., & Abreu, A. (2015). Transcutaneous tibial nerve stimulation in the treatment of lower urinary tract symptoms and its impact on health-related quality of life in patients with Parkinson disease: a randomized controlled trial. Journal of Wound Ostomy & Continence Nursing, 42(1), 94-99.
2. Schneider, M. P., Gross, T., Bachmann, L. M., Blok, B. F., Castro-Diaz, D., Del Popolo, G., ... & Kessler, T. M. (2015). Tibial nerve stimulation for treating neurogenic lower urinary tract dysfunction: a systematic review. European urology, 68(5), 859-867.
“What's wrong with children?”
As pelvic health physical therapists we take care of people suffering from bladder and bowel incontinence and/or dysfunction as well as pre-natal/ post-partum back pain, weak core muscles and pelvic pain. I was approached over 30 years ago by a urologist to take care of his pediatric patients. My reply: “What’s wrong with children?” It’s been a whirlwind of learning since that day!
Pediatric pelvic floor dysfunction is common and can have significant consequences on quality of life for the child and the family, as well as negative health consequences to the lower urinary tract if left untreated.
 According to the National Institute of Diabetes and Digestive and Kidney Diseases, by 5 years of age, over 90% of children have daytime bladder control (NIDDK, 2013) What is life like for the other 10% who experience urinary leakage during the day?
According to the National Institute of Diabetes and Digestive and Kidney Diseases, by 5 years of age, over 90% of children have daytime bladder control (NIDDK, 2013) What is life like for the other 10% who experience urinary leakage during the day?
Bed-wetting is also a pediatric issue with significant negative quality of life impact for both children and their caregivers, with as much as 30% of 4-year-olds experiencing urinary leakage at night (Neveus, 2010). Children who experience anxiety-causing events may have a higher risk of developing urinary incontinence, and in turn, having incontinence causes considerable stress and anxiety for children (Austin, 2014; Neveus, 2010).
Additionally, bowel dysfunction, such as constipation, is a contributor to urinary leakage or urgency. With nearly 5% of pediatric office visits occurring for constipation (Thibodeau 2013, NIDDK, 2013), the need to address these issues is great! And, since pediatric bladder and bowel dysfunction can persist into adulthood, we must direct attention to the pediatric population to improve the health of all our patients.
Children suffer from many diagnoses that affect the pelvic floor including (Austin et al, 2014);
- Voiding dysfunction
- Enuresis (Bedwetting)
- Daytime urinary incontinence
- Urinary urgency and frequency
- Vesicoureteral reflux (Backflow of urine into the kidney)
- Pelvic pain (yes pelvic pain!)
The most common diagnoses I treat are voiding dysfunction and constipation. Pediatric voiding dysfunction is defined as involuntary and intermittent contraction or failure to relax the urethral muscles while emptying the bladder. (Austin et al, 2014); The dysfunctional voiding can present with variable symptoms including urinary urgency, urinary frequency, incontinence, urinary tract infections, and vesicoureteral reflux. Frequently, constipation is a culprit or cause. (Austin et al, 2014; Hodges S. 2012); Managing constipation can have a very positive effect on voiding dysfunction.
“What do we do to teach the pelvic floor (Kegel) muscles to work?”
Common questions I am asked include:
- Can I use biofeedback with children?
- Do we complete internal assessments on pediatric patients?
- How do we teach kids so they can understand?
- Do kids have the ability to learn strengthening versus relaxation?
- How do you teach a child to become aware of their pelvic floor and coordinate it?
If you have pondered these questions, let’s delve in! I see children as young as 4 who have been able to master biofeedback and recite back to me how their pelvic floor works with bowel and bladder function! Children are so eager to please and they love working with animated biofeedback sessions. The research supports the potential benefit of biofeedback training for children with pelvic floor dysfunction (DePaepe et al. 2002, Kaye 2008, Kajbafzadeh 2011, Fazeli 2014). The children are engaged and learn how to isolate their pelvic floor muscles (PFM) through positioning and breathing. The exercises are fun and easy to do. We also incorporate the core! What a wonderful opportunity we have to educate the younger population on these vital muscles as well as proper diet and bowel/bladder habits!
It is not typical to complete an internal pelvic muscle assessment on children, as this would not be appropriate.
“How do I treat it?”
In the literature on pediatric bowel and bladder dysfunction you will often come across the word "Urotherapy". It is, by definition, a conservative management-based program used to treat lower urinary tract (LUT) dysfunction. (Austin 2014)
Basic Urotherapy includes education on the anatomy, behavior modifications including fluid intake, timed or scheduled voids, toileting postures and avoidance of holding maneuvers, diet, avoiding bladder irritants and constipation. Parents are often not aware of their children’s voiding habits once they are cleared from diaper duty after successful potty training occurs.
Urotherapy alone can be helpful however a recent study (Chase, 2010) demonstrated a much greater improvement in those patients who received pelvic floor muscle training as compared to Urotherapy alone.
The International Children’s Continence Society (ICCS) has now expanded the definition of Urotherapy to include Specific Urotherapy (Austin et al, 2014). This includes biofeedback of the pelvic floor muscles by a trained professional who can teach the child how to alter pelvic floor muscle activity specifically for voiding. Cognitive behavioral therapy and psychotherapy are also important and can be a needed in combination with biofeedback in specific cases.
As you can see, PFM exercise combined with Urotherapy is a safe, inexpensive, and effective treatment option for children with pediatric voiding dysfunction.
Do bladder and bowel problems cause psychological problems or is the reverse true?
When we think of pediatric bowel and bladder issues, we primarily focus on what is happening to cause the bowel or bladder leakage and treat it accordingly. It is imperative to teach a child that she/he did not have an “accident”, but their bladder or bowel had a leak. It makes the incident a physiological problem and not something they did. See my blog post on “Accident” for more information.
It is not always apparent how much the child is suffering from issues with self-esteem, embarrassment, internalizing behaviors, externalizing behaviors or oppositional defiant disorders. Dr. Hinman recognized theses issues years ago (1986) and commented that voiding dysfunctions might cause psychological disturbances rather than the reverse being true. Dr. Rushton in 1995 wrote that although a high number of children with enuresis are maladjusted and exhibit measurable behavioral symptoms, only a small percentage have significant underlying psychopathology. In other more recent studies (Joinson et al. 2006a, 2006b, 2008, Kodman-Jones et al, 2001) it was noted that elevated psychological test scores returned to normal after the urologic problem was cured.
I frequently get testimonials from my patients. I would say the common denominator is the child and/or caregivers report that the child is “much better adjusted,” “happier”, “come out of his shell”, “more outgoing”, “making friends.” As a side note -- they’re happy they don’t leak anymore.
You can learn more about treating pediatric patients in my courses,
Pediatric Incontinence and Pelvic Floor Dysfunction and Pediatric Functional Gastrointestinal Disorders.
Austin, P., Bauer, S.B., Bower, W., et al. The standardization of terminology of lower urinary tract function in children and adolescence: update report from the standardization committee of the international children’s continence society. J Urol (2014) 191.
Chase J, Austin P, Hoebeke P, McKenna P. The management of dysfunctional voiding in children: a report from the standarisation committee of the international children’s continence society. 2010; J Urol183:1296-1302.
Constipation in Children. (2013)retrieved June 9, 2014 from http://kidney.niddk.nih.gov/kudiseases/pubs/uichildren/index.aspx
DePaepe H., Renson C., Hoebeke P., et al: The role of pelvic- floor therapy in the treatment of lower urinary tract dysfunctions in children. Scan J of Urol and Neph 2002; 36: 260-7.
Farahmand, F., Abedi, A., Esmaeili-dooki, M. R., Jalilian, R., & Tabari, S. M. (2015). Pelvic Floor Muscle Exercise for Paediatric Functional Constipation.Journal of Clinical and Diagnostic Research : JCDR, 9(6), SC16–SC17. http://doi.org/10.7860/JCDR/2015/12726.6036
Fazeli MS, Lin Y, Nikoo N, Jaggumantri S1, Collet JP, Afshar K. Biofeedback for Non-neuropathic daytime voiding disorders in children: A systematic review and meta-analysis of randomized controlled trials. J Urol. 2014 Jul 26. pii: S0022-5347(14)04048-8.
Hinman, F. Nonneurogenic neurogenic bladder (the Hinman Syndrome)-15 years later. J Urol 1986;136, 769-777.
Hodges SJ, Anthony E. Occult megarectum:a commonly unrecognized cause of enuresis. Urology. 2012 Feb;79(2):421-4. doi: 10.1016/j.urology.2011.10.015. Epub 2011 Dec 14.
Hoebeke, P., Walle, J. V., Theunis, M., De Paepe, H., Oosterlinck, W., & Renson, C. Outpatient pelvic-floor therapy in girls with daytime incontinence and dysfunctional voiding. Urology 1996; 48, 923-927.
Joinson, C., Heron, J., von Gontard, A. and the ALSPAC study team: Psychological problems in children with daytime wetting. Pediatrics 2006a; 118, 1985-1993.
Joinson, C., Heron, J., Butler, U., von Gontard, A. and the ALSPAC study team: Psychological differences between children with and without soiling problems. Pediatrics 2006b; 117, 1575-1584.
Joinson, C., Heron, J., von Gontard, A., Butler, R., Golding, J., Emond, A.: Early childhood risk factors associated with daytime wetting and soiling in school-age children. Journal of Pediatric Psychology2008; e-published.
Kajbafzadeh AM, harifi-Rad L, Ghahestani SM, Ahmadi H, Kajbafzadeh M, Mahboubi AH. (2011) Animated biofeedback: an ideal treatment for children with dysfunctional elimination syndrome. J Urol;186, 2379-2385.
Kaye JD, Palmer LS (2008) Animated biofeedback yields more rapid results than nonanimated biofeedback in the treatment of dysfunctional voiding in girls. J Urol 180, 300-305
Kodman-Jones, C., Hawkins, L., Schulman, SL. Behavioral characteristics of children with daytime wetting. J Urol 2001;Dec(6):2392-5.
Neveus, T, Eggert P, Evans J, et al. Evaluation of the treatment for monosymptomatic enuresis: a standarisation document from the international children’s continence society. J Urol 2010; 183: 441-447
Rushton, H. G. Wetting and functional voiding disorders. Urologic Clinics of North America, 1995; 22(1), 75-93.
Seyedian, S. S. L., Sharifi-Rad, L., Ebadi, M., & Kajbafzadeh, A. M. (2014). Combined functional pelvic floor muscle exercises with Swiss ball and urotherapy for management of dysfunctional voiding in children: a randomized clinical trial. European Journal of Pediatrics, 173(10), 1347-1353.
Thibodeau, B. A., Metcalfe, P., Koop, P., & Moore, K. (2013). Urinary incontinence and quality of life in children. Journal of pediatric urology, 9(1), 78-83.
Urinary Incontinence in Children. (2012). Retrieved June 9, 2014 from http://kidney.niddk.nih.gov/kudiseases/pubs/uichildren/index.aspx
Zivkovic V, Lazovic M, Vlajkovic M, Slavkovic A, Dimitrijevic L, Stankovic I, Vacic N. (2012). Diaphragmatic breathing exercises and pelvic floor retraining in children with dysfunctional voiding. European Journal of Physical Rehabilitation Medicine. 48(3):413-21. Epub 2012 Jun 5.
Dustienne Miller MSPT, WCS, CYT is a Herman & Wallace faculty member, owner of Your Pace Yoga, and the author of the course Yoga for Pelvic Pain. Join her in Columbus, OH this April 27-28, to learn how yoga can be used to treat interstitial cystitis/painful bladder syndrome, vulvar pain, coccydynia, hip pain, and pudendal neuralgia. The course is also coming to Manchester, NH September 7-8, 2019, and Buffalo, NY on October 5-6, 2019.
How does a yoga program compare to a strength and stretching program for women with urinary incontinence? Dr. Allison Huang1 et al have published another research study, after publishing a pilot study2 on using group-based yoga programs to decrease urinary incontinence. Well-known yoga teachers Judith Hanson Lasater, PhD, and Leslie Howard created the yoga class and home program structure for this research study and the 2014 pilot study. The yoga program was primarily based on Iyengar yoga, which uses props to modify postures, a slower tempo to increase mindfulness, and pays special attention to alignment.
 To be chosen for this study, women had to be able to walk more than 2 blocks, transfer from supine to standing independently, be at least 50 years of age, and experience stress, urge, or mixed urinary incontinence at least once daily. Participants had to be new to yoga and holding off on clinical treatment for urinary incontinence, including pelvic health occupational and physical therapy.
To be chosen for this study, women had to be able to walk more than 2 blocks, transfer from supine to standing independently, be at least 50 years of age, and experience stress, urge, or mixed urinary incontinence at least once daily. Participants had to be new to yoga and holding off on clinical treatment for urinary incontinence, including pelvic health occupational and physical therapy.
28 women were assigned to the yoga intervention group and 28 women were assigned to the control group. The mean age was 65.4 with the age range of 55-83 years of age.
The control group received bi-weekly group class and home program instruction on stretching and strengthening without pelvic floor muscle cuing or relaxation training.
The yoga program met for group class twice a week for 90 minutes each and practiced at home one hour per week. The control group met twice a week for 90 minutes with a one-hour home program every week. Both groups met for 12 weeks.
Both groups received bladder behavioral retraining informational handouts. The information sheets contained education about urinary incontinence, pelvic floor muscle strengthening exercises, urge suppression strategies, and instructions on timed voiding.
The yoga program included 15 yoga postures: Parsvokonasana (side angle pose), Parsvottasana (intense side stretch pose), Tadasana (mountain pose) Trikonasana (triangle pose), Utkatasana (chair pose), Virabhadrasana 2 (warrior 2 pose), Baddha Konasana (bounded angle pose), Bharadvajasana (seated twist pose), Malasana (squat pose), Salamba Set Bandhasana (supported bridge pose), Supta Baddha Konasana (reclined cobbler’s pose), Supta Padagushthasana (reclined big toe pose), Savasana (corpse pose), Viparita Karani Variation (legs up the wall pose), and Salabhasana (locust pose).
Women in the yoga intervention group reported more than 76% average improvement in total incontinence frequency over the 3-month period. Women in the muscle stretching/strengthening (without pelvic floor muscle cuing and relaxation training) control group reported more than 56% reduction in leakage episodes.
Stress urinary incontinence episodes decreased by an average of 61% in the yoga group and 35% in the control group (P = .045). Episodes of urge incontinence decreased by an average of 30% in the yoga group and 17% in the control group (P = .77).
The take away? We know behavioral techniques have been shown to improve quality of life and decrease frequency and severity of urinary incontinence episodes.3 Couple this with our clinical interventions, and our patients have a way to reinforce the work we do in the clinic by themselves, or socially within their community. Yoga can be another tool in the toolbox for optimizing pelvic health.
1) Diokno AC et al. (2018). Effect of Group-Administered Behavioral Treatment on Urinary Incontinence in Older Women: A Randomized Clinical Trial. JAMA Intern Med.1;178(10):1333-1341. doi: 10.1001/jamainternmed.2018.3766.
2) Huang, Alison J. et al. (2019). A group-based yoga program for urinary incontinence in ambulatory women: feasibility, tolerability, and change in incontinence frequency over 3 months in a single-center randomized trial. American Journal of Obstetrics & Gynecology. 220(1) 87.e1 - 87.e13. doi: 10.1016/j.ajog.2018.10.031
3) Huang, A. J., Jenny, H. E., Chesney, M. A., Schembri, M., & Subak, L. L. (2014). A group-based yoga therapy intervention for urinary incontinence in women: a pilot randomized trial. Female pelvic medicine & reconstructive surgery, 20(3), 147-54.
In this post, we want to give a high-level overview of interstitial cystitis and an introduction to other resources if you’d like to dive deeper into treatment the condition. There’s a printable, patient-friendly version of this overview if you’d like to use it in describing the condition with patients. In addition, you may want to review the 8 Myths of Interstitial Cystitis series and the AUA Guidelines for Interstitial Cystitis.
Definition
Interstitial cystitis is defined as pain or pressure perceived to be related to the urinary bladder, associated with lower urinary tract symptoms of more than six weeks duration, in the absence of infection or other identifiable causes.
Unfortunately, for physicians, pelvic floor dysfunction falls under category of ‘unidentifiable cause.’ Interstitial cystitis is really more of a description of symptoms, rather than a discrete diagnosis, and the condition presents in many different ways.

Symptoms
The hallmarks of interstitial cystitis are pelvic pain, often in the suprapubic area or inner thighs, and urinary urgency and frequency. Other common symptoms include pain with intercourse, nocturia, low back pain, constipation, and urinary retention.
Many patients are surprised to realize that symptoms like painful intercourse, low back pain, and constipation are related to their IC diagnosis. This challenges the misconception that issues are arising solely from the bladder, and is a good way to help patients (and their physicians) understand that IC is about more than just the bladder.
Diagnosis
Interstitial cystitis is fundamentally a diagnosis of exclusion. Most patients suspect a urinary tract infection (UTI) when their symptoms first present. It’s actually common for symptoms to start as the result of a UTI, and simply not resolve once the infection has cleared. Patients are often treated with multiple rounds of antibiotics for these ‘phantom’ UTIs, where cultures have come back negative, before an IC diagnosis is considered.
It’s important for us as physical therapists to be able to share with patients that no testing is required to confirm an IC diagnosis, it can be diagnosed clinically. In practice, a urologist will likely want to conduct a cystoscopy, which can rule out more serious issues like bladder cancer as well as check for Hunner’s lesions (wounds in the bladder that are present in about 10% of IC patients). However, after that, no additional testing is needed. The potassium sensitivity test (PST) was formerly used by some urologists, but it has been shown to be useless diagnostically and extremely painful for patients and is not recommended by the American Urological Association. Urodynamic testing is also often conducted, but again is not necessary to establish an IC diagnosis.
Physical Therapy for IC
According to the American Urological Association, physical therapy is the most proven treatment for interstitial cystitis. It’s given an evidence grade of ‘A’ (the only treatment with that grade) and recommended in the first line of medical treatment.
In controlled clinical trials, manual physical therapy has been shown to benefit up to 85% of both men and women. These trials reported benefits after ten visits of one-hour treatment sessions.
In a study conducted at our clinic , PelvicSanity, we found that physical therapy was able to reduce pain for IC patients from an average of 7.6 (out of 10) before treatment to 2.6 following physical therapy. Similarly, how much their symptoms bothered patients fell from 8.3 to 2.8. More than half of patients reported improvements within the first three visits.
Unfortunately, many patients still aren’t referred to pelvic physical therapy by their physician. More than half of the patients in the study had seen more than 5 physicians before finding pelvic PT, and only 7% of patients felt they had been referred to physical therapy at the appropriate time by their doctor.
Multi-Disciplinary Approach
Patients with interstitial cystitis or pelvic pain always benefit from a multidisciplinary approach to treatment.This can include:
- Stress relief to downregulate the nervous system can decrease symptoms and reduce flares. Gentle exercise, meditation, yoga, deep breathing, or working with a psychologist can all provide benefits for patients.
- Diet and nutrition are important when working with IC patients. There is no formal ‘IC Diet’, but most patients are sensitive to at least a few trigger foods. The gold standard of treatment is an elimination diet, where the common culprits are completely removed from the diet and then added back in one at a time. This identifies which foods are triggers for patients. With nutrition for IC, patients should avoid their personal trigger foods and eat healthy – it doesn’t have to be any more restrictive or complicated.
- Alternative treatments like acupuncture have been shown to reduce pelvic pain in patients, and several supplements have shown benefits in trials or anecdotally among patients.
- Bladder treatments include instillations and nerve stimulation. Some patients may benefit from bladder instillations, but many others find that the process of the instillation actually causes additional symptoms. If instillations are beneficial, patients should be encouraged to address the underlying issues during the reprieve that instillations bring. Percutaneous tibial nerve stimulation or an implanted nerve stimulation device can both be possible treatment options.
- Oral medications can also reduce symptoms, but do not address the underlying cause of symptoms in patients. Medication that dampens the nervous system, often an anti-depressant or similar medication, can reduce pain and hypersensitivity. Anti-inflammatories may be beneficial in lowering inflammation and helping break the cycle of dysfunction-inflammation-pain. Most patients are started on Elmiron®, the only FDA-approved medication for IC; unfortunately, in the most recent clinical trial research Elmiron has been shown to be no more effective than a placebo. If it is effective, it only is beneficial for about one-third of patients, and many won’t be compliant with the drug due to cost and side effects.
 Nicole Cozean, PT, DPT, WCS (www.pelvicsanity.com/about-nicole) is the founder of PelvicSanity physical therapy in Southern California. Name the 2017 PT of the Year by the ICN, she’s the first physical therapist to serve on the Interstitial Cystitis Association’s Board of Directors and the author of the award-winning book The IC Solution (www.pelvicsanity.com/the-ic-solution). She teaches at her alma mater, Chapman University, as well as continuing education through Herman & Wallace. Nicole also founded the Pelvic PT Huddle (www.facebook.com/groups/pelvicpthuddle), an online Facebook group for pelvic PTs to collaborate.
Nicole Cozean, PT, DPT, WCS (www.pelvicsanity.com/about-nicole) is the founder of PelvicSanity physical therapy in Southern California. Name the 2017 PT of the Year by the ICN, she’s the first physical therapist to serve on the Interstitial Cystitis Association’s Board of Directors and the author of the award-winning book The IC Solution (www.pelvicsanity.com/the-ic-solution). She teaches at her alma mater, Chapman University, as well as continuing education through Herman & Wallace. Nicole also founded the Pelvic PT Huddle (www.facebook.com/groups/pelvicpthuddle), an online Facebook group for pelvic PTs to collaborate.
Interstitial Cystitis Course
In our upcoming course for physical therapists in treating interstitial cystitis (April 6-7, 2019 in Princeton, New Jersey), we’ll focus on the most important physical therapy techniques for IC, home stretching and self-care programs, and information to guide patients in creating a holistic treatment plan. The course will delve into how to handle complex IC presentations. It’s a deep dive into the condition, focusing not just on manual treatment techniques but also how to successfully manage an IC patient from beginning to resolution of symptoms.
Additional Resources
- Interstitial Cystitis Overview (printable)
- The Interstitial Cystitis Solution
- Patient groups include the Interstitial Cystitis Association (ICA) (www.ic-help.org) and the IC Network (www.ic-network.com), which both have fantastic resources for patients.
- The AUA Guidelines for IC
- IC Flare-Busting Plan
A question that often comes up in conversation around menopause is that of pelvic health – the effects on bladder, bowel or sexual health…what works, what’s safe, what’s not? Is hormone therapy better, worse or the same in terms of efficacy when compared to pelvic rehab? Do we have a role here?
An awareness of pelvic health issues that arise at menopause was explored in Oskay’s 2005 paper ‘A study on urogenital complaints of postmenopausal women aged 50 and over’ stating ‘…Urinary incontinence and sexual problems, particularly decline in sexual desire, are widespread among postmenopausal women. Frequent urinary tract infections, obesity, chronic constipation and other chronic illnesses seem to be the predictors of UI.’
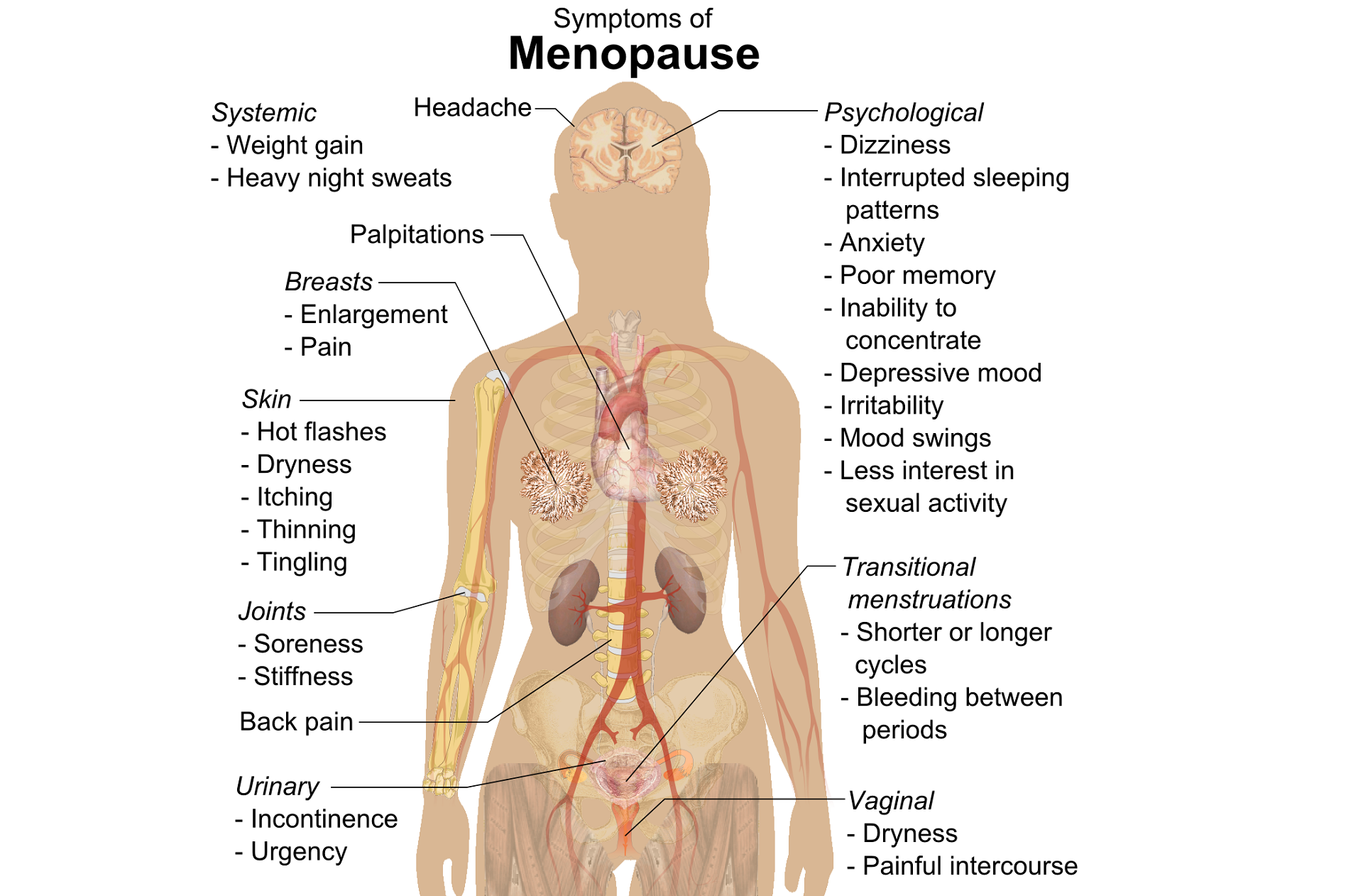 Moller’s 2006 paper explored the link between LUTS (Lower Urinary Tract Symptoms) and sexual activity at midlife: the paper discussed how lower urinary tract symptoms (LUTS) have a profound impact on women’s physical, social, and sexual well being, and confirmed that LUTS are likely to affect sexual activity. However, they also found that conversely, sexual activity may affect the occurrence of LUTS – in their study a questionnaire was sent to 4,000 unselected women aged 40–60 years, and they found that compared to women having sexual relationship, a statistically significant 3 to 6 fold higher prevalence of LUTS was observed in women with no sexual relationship. They also found that women who ceased sexual relationship an increase in the de novo occurrence of most LUTS was observed, concluding that ‘…sexual inactivity may lead to LUTS and vice versa’.
Moller’s 2006 paper explored the link between LUTS (Lower Urinary Tract Symptoms) and sexual activity at midlife: the paper discussed how lower urinary tract symptoms (LUTS) have a profound impact on women’s physical, social, and sexual well being, and confirmed that LUTS are likely to affect sexual activity. However, they also found that conversely, sexual activity may affect the occurrence of LUTS – in their study a questionnaire was sent to 4,000 unselected women aged 40–60 years, and they found that compared to women having sexual relationship, a statistically significant 3 to 6 fold higher prevalence of LUTS was observed in women with no sexual relationship. They also found that women who ceased sexual relationship an increase in the de novo occurrence of most LUTS was observed, concluding that ‘…sexual inactivity may lead to LUTS and vice versa’.
So, who advises women going through menopause about issues such as sexual ergonomics, the use of lubricants or moisturisers, or provide a discussion about the benefits of local topical estrogen? As well as providing a skillset that includes orthopaedic assessment to rule out any musculo-skeletal influences that could be a driver for sexual dysfunction? That would be the pelvic rehab specialist clinician! Tosun et al asked the question ‘Do stages of menopause affect the outcomes of pelvic floor muscle training?’ and the answer in this and other papers was yes; with the research comparing pelvic rehab vs hormone therapy vs a combination approach of pelvic rehab and topical estrogen providing the best outcomes. Nygaard’s paper looked at the ‘Impact of menopausal status on the outcome of pelvic floor physiotherapy in women with urinary incontinence’ and concluded that : ‘…(both pre and postmenopausal women) benefit from motor learning strategies and adopt functional training to improve their urinary symptoms in similar ways, irrespective of hormonal status or HRT and BMI category’.
We must also factor in some of the other health concerns that pelvic health can impact at midlife for women – Brown et al asked the question ‘Urinary incontinence: does it increase risk for falls and fractures?’ They answered their question by concluding that ‘‘… urge incontinence was associated independently with an increased risk of falls and non-spine, nontraumatic fractures in older women. Urinary frequency, nocturia, and rushing to the bathroom to avoid urge incontinent episodes most likely increase the risk of falling, which then results in fractures. Early diagnosis and appropriate treatment of urge incontinence may decrease the risk of fracture.’
If you are interested in learning more about pelvic health, sexual function and bone health at Menopause, consider attending Menopause Rehabilitation and Symptom Management.
Sexual activity and lower urinary tract symptoms’ Møller LA1, Lose G. Int Urogynecol J Pelvic Floor Dysfunct. 2006 Jan;17(1):18-21. Epub 2005 Jul 29.
A study on urogenital complaints of postmenopausal women aged 50 and over. Oskay UY1, Beji NK, Yalcin O. Acta Obstet Gynecol Scand. 2005 Jan;84(1):72-8.
Do stages of menopause affect the outcomes of pelvic floor muscle training? Tosun ÖÇ1, Mutlu EK, Tosun G, Ergenoğlu AM, Yeniel AÖ, Malkoç M, Aşkar N, İtil İM. Menopause. 2015 Feb;22(2):175-84. doi: 10.1097/GME.0000000000000278.
‘Impact of menopausal status on the outcome of pelvic floor physiotherapy in women with urinary incontinence.’ Nygaard CC1, Betschart C, Hafez AA, Lewis E, Chasiotis I, Doumouchtsis SK. Int Urogynecol J. 2013 Dec;24(12):2071-6. doi: 10.1007/s00192-013-2179-7. Epub 2013 Jul 17
Sara Chan Reardon, DPT, WCS, BCB-PMD is a pelvic floor dysfunction specialist practicing in New Orleans, LA. Sara was named the 2008 Section on Women’s Health Research Scholar for her published research on pelvic floor dyssynergia related constipation. She was recognized as an Emerging Leader in 2013 by the American Physical Therapy Association. She served as Treasurer of the APTA’s Section on Women's Health and sat on their Executive Board of Directors from 2012-2015. Today she was kind enough to share a bit about her course Post-Prostatectomy Patient Rehabilitation, which is taking place twice in 2018.
 My name is Sara Reardon, and I teach the Post-Prostatectomy Patient Rehabilitation course, which I wrote and developed in the year 2015. At the time, I had been a pelvic health Physical Therapist for over 10 years. Earlier in my career, I had taken the Pelvic Floor 2A course by Herman and Wallace Institute, which was a fantastic and thorough introduction to treating a male patient.
My name is Sara Reardon, and I teach the Post-Prostatectomy Patient Rehabilitation course, which I wrote and developed in the year 2015. At the time, I had been a pelvic health Physical Therapist for over 10 years. Earlier in my career, I had taken the Pelvic Floor 2A course by Herman and Wallace Institute, which was a fantastic and thorough introduction to treating a male patient.
Over the years, I started seeing more and more men with post-prostatectomy urinary incontinence and erectile dysfunction in my clinic. Urinary incontinence is the most common and costly complication in men following prostate removal surgery, and their quality of life is directly related to their duration of experiencing those symptoms. Evidence supports that pelvic floor muscle training started as soon as possible after surgery can help decrease incontinence and improve quality of life. I enjoyed being able to help men decrease their incontinence and improve their other symptoms after all they had been through following a cancer diagnosis and treatment.
No courses focused specifically on treating post-prostatectomy pelvic floor dysfunction were offered at the time, so I scoured the research, shadowed with physicians, observed surgeries, and attended urology conferences to understand how to effectively treat these individuals. Treating this population of men is fun, fulfilling, and rewarding, and I was inspired to help other pelvic health physical therapists dive deeper as I witnessed the impact pelvic health physical therapy can have on the quality of life of these patients. I love teaching this course, and I am excited to help other pelvic health professionals learn evidence based and effective treatment strategies to help these men navigate their recovery after prostatectomy.
Join Dr. Reardon in Philadelphia, PA on June 2-3, 2018 or in Houston, TX on November 10-11, 2018 to learn evaluation and treatment techniques for men recovering from prostatectomy surgery.
A 2016 study by Kaori et al examined the effect of self administered perineal stimulation for nocturia in elderly women. A prior study using rodents found a soft roller used decreased overactive bladder syndrome (OAB), but a hard roller did not produce the same results. Kaori et al performed a similar study for elderly women in a randomized, placebo controlled, double blind crossover. Participants were 79-89 years old women who applied simulation to perineal skin for 1 minute at bedtime, using either active (soft, sticky elastomer) roller or a placebo (hard polylestrene roller). Participants did a 3-day baseline, followed by 3-day stimulation, then 4 days rest, then other stimuli for 3 days. There were 24 participants, 22 completed the study: 9 with OAB, 13 without OAB. The placement of the roller was not on the skin of the perineal body, but rather on the general peri-anal area with the diagram from the study showing an area just medial to the gluteal crease—where one would find the ischial tuberosity-- and anterior and lateral to the anal sphincter.
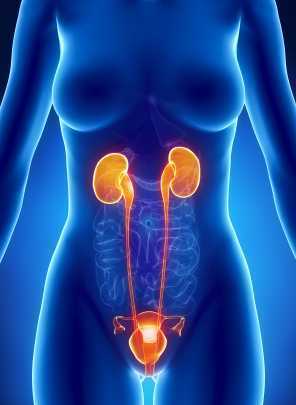 Across the subjects with OAB, change with the elastomer roller (soft and sticky feel) was more statistically significant than with the hard roller. Baseline micturition for the participants was 3.2+/- 1.2 times per night, measured as the number of urination between going to bed and arising. The group as a whole did not have a statistically significant difference, measured by at least one less time arising per night. However, in the OAB group, the difference was significant. The researchers theorized that the soft and sticky texture may induce more firing of somatic afferents nerve fibers.
Across the subjects with OAB, change with the elastomer roller (soft and sticky feel) was more statistically significant than with the hard roller. Baseline micturition for the participants was 3.2+/- 1.2 times per night, measured as the number of urination between going to bed and arising. The group as a whole did not have a statistically significant difference, measured by at least one less time arising per night. However, in the OAB group, the difference was significant. The researchers theorized that the soft and sticky texture may induce more firing of somatic afferents nerve fibers.
The most commonly prescribed treatment for overactive bladder is anticholinergic therapy, but the side effects, including cognitive changes and lack of significant difference from controls, as well as the drying effect of these drugs in a post-menopausal-low-estrogen-pelvis, bring up questions of whether this is the best option in the elderly.(6)
In anesthetized animals, electrical stimulation and noxious stimuli decrease frequency of bladder contractions when applied to the perineal area (3-5). Somatic, afferent nerve stimuli (those theorized to be active with the soft roller) are used to treat OAB by modalities such as acupuncture and transcutaneous electrical stimulation to the perineum (2). So, stimulation of somatic visceral afferent nerves in the perineal region seems to have an effect on the bladder. However, with manual therapies, it seems we can also affect the somatic or visceral afferents. Essentially, visceral afferents convey information to the central nervous system about local changes in chemical and mechanical environments of a number of organ systems(7). Doing manual therapy between the urethral and bladder fascia would also theoretically cause stimulation of the visceral afferents to the central nervous system about that organ (bladder).
In our pelvic floor intro class (Pelvic Floor Level 1) at Herman Wallace, we discuss the role of Bradley’s neurology loop 3 and the inverse relationship between pelvic floor contraction (lifting the perineal area) and the bladder. One suppression technique we discuss is the contraction of the pelvic floor to quiet or inhibit bladder activity in the bladder retraining program. Bladder retraining has evidence level A (strong) for improving urgency and frequency with overactive bladder.
Clinicians who are ready to raise their manual game may try using the skills of prior series courses and adding the sophistication of manual techniques in the abdomen and pelvis to increase afferent firing in patients with OAB, as well as freeing up any fascial restrictions that may be interfering with full bladder excursion.
In the newly written Capstone course, we combine the prior level of education from the pelvic series (bladder strategies) with manual techniques to address the endopelvic fascia at the bladder base, in the fascial articulations along the perineum, and along its attachments to the coccyx, as well as combining internal work with sacral techniques to facilitate S234 afferents for bladder control. We discuss studies, such as this one, to explore advanced concepts of bladder and urethral fascial mechanics and neural entrapment affecting the bladder. We move out of the pelvic muscle and into the fascial contents of the abdominopelvic region, to allow such firing of the somatic afferents. And the perineal stimulation? We have an entire lab for perineal tissue and its effect on pelvic function. Physical therapists can manually address the perineum, urethral and bladder fascia with Capstone techniques. With such intervention, we get more CNS communication.
So, what about the roller? Well, the soft roller created change in rodents in a couple of studies. (Sato 2010). In this human study, it helped with OAB. Certainly, manual therapies in the region of the endopelvic fascia and suprapubic region may be of help for also stimulating the visceral afferents. Also, it could be worth it to have a high fall risk elderly patient with OAB type nocturia follow up your treatments with one minute of soft washcloth stroking in the area of the perineum for one minute at bedtime to see if it helps decrease the number of voids on a night time bladder diary.
Nari Clemons, PT, PRPC is a Herman & Wallace faculty member who helped author the Pelvic Floor Series Capstone: Advanced Topics in Pelvic Rehab course. She is also the creator and instructor of Pelvic Nerve Manual Assessment and Treatment.
Main study: PLoS One. 2016 Mar 22;11(3):e0151726. doi: 10.1371/journal.pone.0151726. eCollection 2016.Effects of a Gentle, Self-Administered Stimulation of Perineal Skin for Nocturia in Elderly Women: A Randomized, Placebo-Controlled, Double-Blind Crossover Trial.Iimura K1,2, Watanabe N2, Masunaga K3, Miyazaki S1,2,4, Hotta H2, Kim H5, Hisajima T1,4, Takahashi H1,4, Kasuya Y3.
2. Exp Ther Med. 2013 Sep;6(3):773-780. Epub 2013 Jul 9., Acupuncture for the treatment of urinary incontinence: A review of randomized controlled trials.Paik SH1, Han SR, Kwon OJ, Ahn YM, Lee BC, Ahn SY.
3. Guo ZF. Transcutaneious electrical nerve stimulation in the treatment of patients with poststroke urinary incontinence. Clin Interv Aging. 2014; 851-6.
4. Sato A, The impact of somatosensory input on autonomic functions. Reve Physiol Biochem Pharmacol. 1997;130;1-328
5. Sato A. Mechanism of the reflex inhibition of micturition conractions of the urinary bladder elicited by acupuncture-like stimulation in anesthetized rats. Neurosci res. 1992 15:189-98
6). Effects of a Gentle, Self-Administered Stimulation of Perineal Skin for Nocturia in Elderly Women: A Randomized, Placebo-Controlled, Double-Blind Crossover Trial. Iimura K, Watanabe N, Masunaga K, Miyazaki S, Hotta H, Kim H, Hisajima T, Takahashi H, Kasuya Y. PLoS One. 2016 Mar 22;11(3):e0151726. doi: 10.1371/journal.pone.0151726. eCollection 2016.
7) John C. Longhurst, Liang-Wu Fu, in Primer on the Autonomic Nervous System (Third Edition), 2012